ជំងឺកូរ៉ូណាវីរុស២០១៩
| អត្ថបទនេះត្រូវការបកប្រែទៅជាភាសាខ្មែរ។ អត្ថបទនេះត្រូវបានសរសេរជាភាសាផ្សេង ដែលមិនមែនជាភាសាខ្មែរ។ បើសិនជាអត្ថបទទុកសម្រាប់អ្នកអានមកពី សហគមន៍នៃភាសាមួយនេះ វាគួរតែចែកចាយទៅវិគីភីឌាជាភាសានោះ។ សូមមើល បញ្ជីនៃគម្រោងវិគីភីឌាទាំងអស់។ សូមមើល ច្រកចូលអត្ថបទនេះ លើក្រុមទំព័រដែលត្រូវការបំណកប្រែទៅជាភាសាខ្មែរ ដើម្បីពិភាក្សា ។ ប្រសិនបើ អត្ថបទមិនត្រូវបានសរសេរជាភាសាខ្មែរឡើងវិញទេ ក្នុងរយៈពេលពីរសប្ដាហ៍ទៀត អត្ថបទនឹងត្រូវចុះបញ្ជីដើម្បីលុបចោល និង/ឬ ប្ដូរវាទៅកាន់វិគីភីឌាជាភាសាដើមរបស់វាវិញ ។ បើសិនជាលោកអ្នក គ្រាន់តែចង់បិទស្លាកទំព័រនេះត្រូវការបំណកប្រែ សូមបញ្ចូល {{អាទេស.:ត្រូវការបកប្រែ | ទំ. = ជំងឺកូរ៉ូណាវីរុស២០១៩ | ភាសា = មិនស្គាល់ | ផ្ដល់យោបល់ = }} ~~~~ទៅខាងក្រោម នៃផ្នែក នៃក្រុមទំព័រនេះត្រូវការបំណកប្រែទៅជាភាសាខ្មែរ ។ |
អត្ថបទនេះ ហួសសម័យហើយ។ សូមបន្ទាន់សម័យអត្ថបទនេះ ដើម្បីឆ្លុះបង្ហាញឱ្យឃើញនូវព្រឹត្តិការណ៍ថ្មី ឬ ព័ត៌មានដែលបានចេញថ្មីៗនេះ។ |
| ជំងឺកូរ៉ូណាវីរុស២០១៩ (កូវីដ-១៩) | |
|---|---|
| ឈ្មោះផ្សេងៗ | |
 | |
| រោគសញ្ញាកូវីដ-១៩ | |
| ឯកទេសភាព | ជំងឺបង្ករោគ |
| រោគសញ្ញា | ឡើងកម្តៅ, ក្អក, ពិបាកដកដង្ហើម, បាត់បង់ឃានវិញ្ញាណ (ក្លិន)[៣][៤][៥] |
| ដង្ហកធ្វើទុក្ខ | រោគរលាកសួត, ពិសឈាមវីរុសviral sepsis, acute respiratory distress syndrome, ការខ្សោយតំរងនោមkidney failure, សមាការព្រលែងកោសិការចលកម្មcytokine release syndrome |
| អាយុចេញរោគសញ្ញា | ២–១៤ ថ្ងៃ (ជាធម្មតា ៥) ពីការឆ្លង |
| ហេតុបង្ក | កូរ៉ូណាវីរុសសមាការផ្លូវដង្ហើមស្រួចស្រាវធ្ងន់ធ្ងរ២ (សារស៍-កូវ-២) |
| កត្តាហានិភ័យ | ធ្វើដំណើរ, ទៅកន្លែងណាដែលងាយប្រថុយនឹងវីរុស |
| វិធីធ្វើរោគវិនិច្ឆ័យ | សំណាក rRT-PCR testing, ការឆ្លុះCT scan |
| ការបង្ការ | ការលាងដៃ, បិទមុខ, ចត្តាឡីស័ក (នៅដោយឡែក)[៦] |
| ការព្យាបាល | តាមរោគសញ្ញាSymptomatic និងand ឧបត្ថម្ភsupportive |
| ភាពញឹកញាប់ | ១០៤៣៣៣៨៧៨[៧] confirmed cases |
| មរណភាព | ២២៦៥៥៥៩ (២% of confirmed cases)[៧] |

ជំងឺវីរុសកូរ៉ូណាឆ្នាំ២០១៩ (កូវីដ-១៩) គឺជាជំងឺឆ្លងដែលបង្កដោយកូរ៉ូណាវីរុសសមាការផ្លូវដង្ហើមស្រួចស្រាវធ្ងន់ធ្ងរ២ (សារ្ស-កូវ-២).[៨] កូវីដ-១៩ ត្រូវបានគេរកឃើញដំបូងក្នុងខែធ្នូ ឆ្នាំ២០១៩ នៅក្នុងទីក្រុងអ៊ូហាន, រដ្ឋធានីនៃខេត្តហ៊ូប៉ីរបស់ប្រទេសចិនហើយចាប់តាំងពីពេលនោះមកវាបានរីករាលដាលទៅជុំវិញពិភពលោកហើយបន្តក្លាយជាជំងឺរាតត្បាតសកល។[៩][១០] គិតត្រឹមថ្ងៃ ៤ កុម្ភៈ ២០២១, ករណីប្រមាណ ១០៤ million ករណីត្រូវបានគេរកឃើញនៅតាមប្រទេសនិងដែនដីចំនួន ១៩២ ដែលជាលទ្ធផលមនុស្សប្រមាណជាង ២២៦០០០០ នាក់ត្រូវបានបាត់បង់ជីវិត។ ចំណែកឯអ្នកជាសះស្បើយមានចំនួនជាង ៥៧៩០០០០០ នាក់។[៧]
រោគសញ្ញាទូទៅរួមមានគ្រុនក្តៅកំដៅចាប់ពី ៣៧,៥ុអង្សារឡើងទៅ, ក្អក,ឈឺបំពង់ក, ឆាប់អស់កម្លាំង, ការពិបាកដកដង្ហើម, និងបាត់បង់ឃាណវិញ្ញាណ និងជីវ្ហាវិញ្ញាណ។[៤][១១][១២] រោគសញ្ញានៃករណីកូវីដ-១៩ ភាគច្រើនគឺបែបលក្ខណៈធន់ស្រាល, ប៉ុន្តែខ្លះអាចវិវត្តទៅជារោគរលាកសួត, សរីរាង្គមួយចំនួនឈប់ដំណើរការ, ឫកកសរសៃឈាមជាដើម។[៩][១៣][១៤] រយៈវេលានៃរោគសញ្ញានឹងចាប់ផ្តើមមាននៅថ្ងៃទីប្រាំបន្ទាប់ពីបានប៉ះជាមួយនឹងវីរុសប៉ុន្តែពេលខ្លះវានឹងចំណាយពេលចាប់ពី ២ ថ្ងៃរហូតដល់ទៅ ១៤ ថ្ងៃឯណោះ។[៤][១៥]
The virus is primarily spread between people during close contact,[lower-alpha ១] often via small droplets produced by coughing,[lower-alpha ២] sneezing, and talking.[៥][១៦][១៨] The droplets usually fall to the ground or onto surfaces rather than remaining in the air over long distances.[៥] People may also become infected by touching a contaminated surface and then touching their face.[៥][១៦] On surfaces, the amount of virus declines over time until it is insufficient to remain infectious, but it may be detected for hours or days.[៥][១៨][១៩] It is most contagious during the first three days after the onset of symptoms, although spread may be possible before symptoms appear and in later stages of the disease.[២០] The standard method of diagnosis is by real-time reverse transcription polymerase chain reaction (rRT-PCR) from a nasopharyngeal swab.[២១] Chest CT imaging may also be helpful for diagnosis in individuals where there is a high suspicion of infection based on symptoms and risk factors; however, guidelines do not recommend using it for routine screening.[២២][២៣]
Recommended measures to prevent infection include frequent hand washing, maintaining physical distance from others (especially from those with symptoms), quarantine, covering coughs, and keeping unwashed hands away from the face.[២៤][២៥][៦] In addition, the use of a face covering is recommended for those who suspect they have the virus and their caregivers.[២៦][២៧] Recommendations for face covering use by the general public vary, with some authorities recommending, some recommending against, and others requiring their use.[២៧][២៨][២៩] There is limited evidence for or against the use of masks (medical or other) in healthy individuals in the wider community.[៥]
According to the World Health Organization, there are no available vaccines nor specific antiviral treatments for COVID-19.[៥] On 1 May 2020, the United States gave Emergency Use Authorization to the antiviral remdesivir for people hospitalized with severe COVID-19.[៣០] Management involves the treatment of symptoms, supportive care, isolation, and experimental measures.[៣១] The World Health Organization (WHO) declared the COVID-19 outbreak a Public Health Emergency of International Concern (PHEIC)[៣២][៣៣] on 30 January 2020 and a pandemic on 11 March 2020.[១០] Local transmission of the disease has occurred in most countries across all six WHO regions.[៣៤]
អាការៈ និង រោគសញ្ញា
[កែប្រែ]| Symptom | Range |
|---|---|
| Fever | 83–99% |
| Cough | 59–82% |
| Loss of appetite | 40–84% |
| Fatigue | 44–70% |
| Shortness of breath | 31–40% |
| Coughing up sputum | 28–33% |
| Muscle aches and pains | 11–35% |
Fever is the most common symptom, although some older people and those with other health problems experience fever later in the disease.[៣][៣៥] In one study, 44% of people had fever when they presented to the hospital, while 89% went on to develop fever at some point during their hospitalization.[៣][៣៦]
Other common symptoms include cough, loss of appetite, fatigue, shortness of breath, sputum production, and muscle and joint pains.[៣][៤][១][៣៧] Symptoms such as nausea, vomiting, and diarrhoea have been observed in varying percentages.[៣៨][៣៩][៤០] Less common symptoms include sneezing, runny nose, or sore throat.[៤១]
Some cases in China initially presented with only chest tightness and palpitations.[៤២]
A decreased sense of smell or disturbances in taste may occur.[៤៣][៤៤] Loss of smell was a presenting symptom in 30% of confirmed cases in South Korea.[១២][៤៥]
As is common with infections, there is a delay between the moment a person is first infected and the time he or she develops symptoms. This is called the incubation period. The incubation period for COVID‑19 is typically five to six days but may range from two to 14 days,[៤៦][៤៧] although 97.5% of people who develop symptoms will do so within 11.5 days of infection.[៤៨]
A minority of cases do not develop noticeable symptoms at any point in time.[៤៩][៥០] These asymptomatic carriers tend not to get tested, and their role in transmission is not yet fully known.[៥១][៥២] However, preliminary evidence suggests they may contribute to the spread of the disease.[៥៣][៥៤] In March 2020, the Korea Centers for Disease Control and Prevention (KCDC) reported that 20% of confirmed cases remained asymptomatic during their hospital stay.[៥៤][៥៥]
Complications
[កែប្រែ]In some, the disease may progress to pneumonia, multi-organ failure, and death.[៩] Neurological manifestations include [kikilu[seizures]], stroke, encephalitis, and Guillain–Barré syndrome.[៥៦] Cardiovascular-related complications may include heart failure, irregular electrical activity, blood clots, and heart inflammation.[៥៧]
In some people, COVID‑19 may affect the lungs causing pneumonia. In those most severely affected, COVID-19 may rapidly progress to acute respiratory distress syndrome (ARDS) causing respiratory failure, septic shock, or multi-organ failure.[៥៨][៥៩] Complications associated with COVID‑19 include sepsis, abnormal clotting, and damage to the heart, kidneys, and liver. Clotting abnormalities, specifically an increase in prothrombin time, have been described in 6% of those admitted to hospital with COVID-19, while abnormal kidney function is seen in 4% of this group.[៦០] Approximately 20-30% of people who present with COVID‑19 demonstrate elevated liver enzymes (transaminases).[៦១] Liver injury as shown by blood markers of liver damage is frequently seen in severe cases.[៦២]
Cause
[កែប្រែ]ការចំលង
[កែប្រែ]
COVID-19 is a new disease, and the ways it spreads between people are under investigation, including: the role of small droplets, the extent and how it may be transmitted through air, and how long the virus remains infectious on surfaces.[៥][១៦][១៨] The disease is spread during close contact, often by small droplets produced during coughing, sneezing, or talking.[៥][១៨] During close contact, (1 to 2 metres, 3 to 6 feet), people catch the disease after breathing in contaminated droplets that were exhaled by infected people. However, the droplets are relatively heavy and usually fall to the ground or surfaces, as opposed to being infectious over large distances.[៥][១៨]
After the droplets fall to floors or surfaces, they still can infect other people, if they touch contaminated surfaces and then their eyes, nose or mouth with unwashed hands.[៥] On surfaces the amount of active virus decreases over time until it can no longer cause infection.[១៨] However, experimentally, the virus can survive on various surfaces for some time, (for example copper or cardboard for a few hours, and plastic or steel for a few days).[១៨][៦៣] Surfaces are easily decontaminated with household disinfectants which kill the virus outside the human body or on the hands.[៥] Disinfectants or bleach are not a treatment for COVID-19, and cause health problems when not used properly, such as inside the human body.[៦៤]
Sputum and saliva carry large amounts of virus.[៦៥][៥][១៦][១៨] Some medical procedures may result in the virus being transmitted easier than normal for such small droplets, known as airborne transmission.[៥][១៨]
The virus is most contagious during the first three days after onset of symptoms, although spread is known to occur up to two days before symptoms appear (presymptomatic transmission) and in later stages of the disease.[១៦][១៨][៦៦][៦៧] Some people have been infected and recovered without showing symptoms, but uncertainties remain in terms of asymptomatic transmission.[១៨]
Although COVID-19 is not a sexually transmitted infection, kissing, intimate contact, and faecal oral routes are suspected to transmit the virus.[៦៨][៦៩]
វីរុសវិទ្យា
[កែប្រែ]
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a novel severe acute respiratory syndrome coronavirus, first isolated from three people with pneumonia connected to the cluster of acute respiratory illness cases in Wuhan.[៧០] All features of the novel SARS-CoV-2 virus occur in related coronaviruses in nature.[៧១] Outside the human body, the virus is killed by household soap, which bursts its protective bubble.[២២]
SARS-CoV-2 is closely related to the original SARS-CoV.[៧២] It is thought to have a zoonotic origin. Genetic analysis has revealed that the coronavirus genetically clusters with the genus Betacoronavirus, in subgenus Sarbecovirus (lineage B) together with two bat-derived strains. It is 96% identical at the whole genome level to other bat coronavirus samples (BatCov RaTG13).[៤១] In February 2020, Chinese researchers found that there is only one amino acid difference in the binding domain of the S protein between the coronaviruses from pangolins and those from humans; however, whole-genome comparison to date found that at most 92% of genetic material was shared between pangolin coronavirus and SARS-CoV-2, which is insufficient to prove pangolins to be the intermediate host.[៧៣]
Pathophysiology
[កែប្រែ]The lungs are the organs most affected by COVID‑19 because the virus accesses host cells via the enzyme angiotensin-converting enzyme 2 (ACE2), which is most abundant in type II alveolar cells of the lungs. The virus uses a special surface glycoprotein called a "spike" (peplomer) to connect to ACE2 and enter the host cell.[៧៤] The density of ACE2 in each tissue correlates with the severity of the disease in that tissue and some have suggested that decreasing ACE2 activity might be protective,[៧៥][៧៦] though another view is that increasing ACE2 using angiotensin II receptor blocker medications could be protective and these hypotheses need to be tested.[៧៧] As the alveolar disease progresses, respiratory failure might develop and death may follow.[៧៦]
SARS-CoV-2 may also cause respiratory failure through affecting the brainstem as other coronaviruses have been found to invade the central nervous system (CNS). While virus has been detected in cerebrospinal fluid of autopsies, the exact mechanism by which it invades the CNS remains unclear and may first involve invasion of peripheral nerves given the low levels of ACE2 in the brain.[៧៨][៧៩]
The virus also affects gastrointestinal organs as ACE2 is abundantly expressed in the glandular cells of gastric, duodenal and rectal epithelium[៨០] as well as endothelial cells and enterocytes of the small intestine.[៨១]
The virus can cause acute myocardial injury and chronic damage to the cardiovascular system.[៨២] An acute cardiac injury was found in 12% of infected people admitted to the hospital in Wuhan, China,[៣៩] and is more frequent in severe disease.[៨៣] Rates of cardiovascular symptoms are high, owing to the systemic inflammatory response and immune system disorders during disease progression, but acute myocardial injuries may also be related to ACE2 receptors in the heart.[៨២] ACE2 receptors are highly expressed in the heart and are involved in heart function.[៨២][៨៤] A high incidence of thrombosis (31%) and venous thromboembolism (25%) have been found in ICU patients with COVID‑19 infections and may be related to poor prognosis.[៨៥][៨៦] Blood vessel dysfunction and clot formation (as suggested by high D-dimer levels) are thought to play a significant role in mortality, incidences of clots leading to pulmonary embolisms, and ischaemic events within the brain have been noted as complications leading to death in patients infected with SARS-CoV-2. Infection appears to set off a chain of vasoconstrictive responses within the body, constriction of blood vessels within the pulmonary circulation has also been posited as a mechanism in which oxygenation decreases alongside the presentation of viral pneumonia.[៨៧]
Another common cause of death is complications related to the kidneys[៨៧]—SARS-CoV-2 directly infects kidney cells, as confirmed in post-mortem studies. Acute kidney injury is a common complication and cause of death; this is more significant in patients with already compromised kidney function, especially in people with pre-existing chronic conditions such as hypertension and diabetes which specifically cause nephropathy in the long run.[៨៨]
Autopsies of people who died of COVID‑19 have found diffuse alveolar damage (DAD), and lymphocyte-containing inflammatory infiltrates within the lung.[៨៩]
Immunopathology
[កែប្រែ]Although SARS-COV-2 has a tropism for ACE2-expressing epithelial cells of the respiratory tract, patients with severe COVID‑19 have symptoms of systemic hyperinflammation. Clinical laboratory findings of elevated IL-2, IL-7, IL-6, granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon-γ inducible protein 10 (IP-10), monocyte chemoattractant protein 1 (MCP-1), macrophage inflammatory protein 1-α (MIP-1α), and tumour necrosis factor-α (TNF-α) indicative of cytokine release syndrome (CRS) suggest an underlying immunopathology.[៣៩]
Additionally, people with COVID‑19 and acute respiratory distress syndrome (ARDS) have classical serum biomarkers of CRS, including elevated C-reactive protein (CRP), lactate dehydrogenase (LDH), D-dimer, and ferritin.[៩០]
Systemic inflammation results in vasodilation, allowing inflammatory lymphocytic and monocytic infiltration of the lung and the heart. In particular, pathogenic GM-CSF-secreting T-cells were shown to correlate with the recruitment of inflammatory IL-6-secreting monocytes and severe lung pathology in COVID‑19 patients.[៩១] Lymphocytic infiltrates have also been reported at autopsy.[៨៩]
Diagnosis
[កែប្រែ]

The WHO has published several testing protocols for the disease.[៩៣] The standard method of testing is real-time reverse transcription polymerase chain reaction (rRT-PCR).[៩៤] The test is typically done on respiratory samples obtained by a nasopharyngeal swab; however, a nasal swab or sputum sample may also be used.[២១][៩៥] Results are generally available within a few hours to two days.[៩៦][៩៧] Blood tests can be used, but these require two blood samples taken two weeks apart, and the results have little immediate value.[៩៨] Chinese scientists were able to isolate a strain of the coronavirus and publish the genetic sequence so laboratories across the world could independently develop polymerase chain reaction (PCR) tests to detect infection by the virus.[៩][៩៩][១០០] As of 4 មេសា 2020[update], antibody tests (which may detect active infections and whether a person had been infected in the past) were in development, but not yet widely used.[១០១][១០២][១០៣] The Chinese experience with testing has shown the accuracy is only 60 to 70%.[១០៤] The FDA in the United States approved the first point-of-care test on 21 March 2020 for use at the end of that month.[១០៥]
Diagnostic guidelines released by Zhongnan Hospital of Wuhan University suggested methods for detecting infections based upon clinical features and epidemiological risk. These involved identifying people who had at least two of the following symptoms in addition to a history of travel to Wuhan or contact with other infected people: fever, imaging features of pneumonia, normal or reduced white blood cell count, or reduced lymphocyte count.[១០៦]
A study asked hospitalised COVID‑19 patients to cough into a sterile container, thus producing a saliva sample, and detected the virus in eleven of twelve patients using RT-PCR. This technique has the potential of being quicker than a swab and involving less risk to health care workers (collection at home or in the car).[៦៥]
Along with laboratory testing, chest CT scans may be helpful to diagnose COVID-19 in individuals with a high clinical suspicion of infection but are not recommended for routine screening.[២២][២៣] Bilateral multilobar ground-glass opacities with a peripheral, asymmetric, and posterior distribution are common in early infection.[២២] Subpleural dominance, crazy paving (lobular septal thickening with variable alveolar filling), and consolidation may appear as the disease progresses.[២២][១០៧]
In late 2019, WHO assigned the emergency ICD-10 disease codes U07.1 for deaths from lab-confirmed SARS-CoV-2 infection and U07.2 for deaths from clinically or epidemiologically diagnosed COVID‑19 without lab-confirmed SARS-CoV-2 infection.[១០៨]
រោគវិទ្យា
[កែប្រែ]Few data are available about microscopic lesions and the pathophysiology of COVID‑19.[១០៩][១១០] The main pathological findings at autopsy are:
- Macroscopy: pleurisy, pericarditis, lung consolidation and pulmonary oedema
- Four types of severity of viral pneumonia can be observed:
- minor pneumonia: minor serous exudation, minor fibrin exudation
- mild pneumonia: pulmonary oedema, pneumocyte hyperplasia, large atypical pneumocytes, interstitial inflammation with lymphocytic infiltration and multinucleated giant cell formation
- severe pneumonia: diffuse alveolar damage (DAD) with diffuse alveolar exudates. DAD is the cause of acute respiratory distress syndrome (ARDS) and severe hypoxemia.
- healing pneumonia: organisation of exudates in alveolar cavities and pulmonary interstitial fibrosis
- plasmocytosis in BAL[១១១]
- Blood: disseminated intravascular coagulation (DIC);[១១២] leukoerythroblastic reaction[១១៣]
- Liver: microvesicular steatosis
ការបង្ការ
[កែប្រែ]
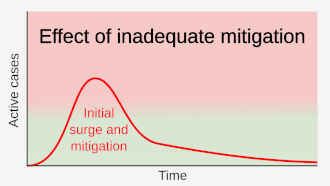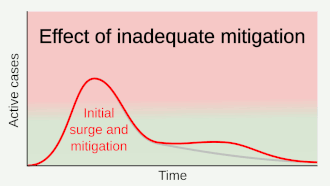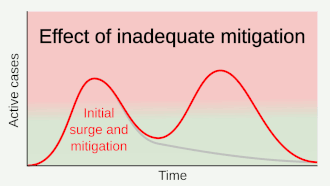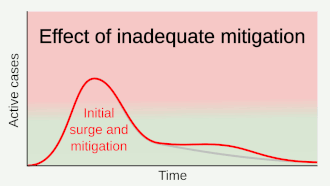
Preventive measures to reduce the chances of infection include staying at home, avoiding crowded places, keeping distance from others, washing hands with soap and water often and for at least 20 seconds, practising good respiratory hygiene, and avoiding touching the eyes, nose, or mouth with unwashed hands.[១១៩][១២០][១២១] The CDC recommends covering the mouth and nose with a tissue when coughing or sneezing and recommends using the inside of the elbow if no tissue is available.[១១៩] Proper hand hygiene after any cough or sneeze is encouraged.[១១៩] The CDC has recommended the use of cloth face coverings in public settings where other social distancing measures are difficult to maintain, in part to limit transmission by asymptomatic individuals.[១២២] The U.S. National Institutes of Health guidelines do not recommend any medication for prevention of COVID‑19, before or after exposure to the SARS-CoV-2 virus, outside of the setting of a clinical trial.[១២៣]
Social distancing strategies aim to reduce contact of infected persons with large groups by closing schools and workplaces, restricting travel, and cancelling large public gatherings.[១២៤] Distancing guidelines also include that people stay at least 6 feet (1.8 m) apart.[១២៥] There is no medication known to be effective at preventing COVID‑19.[១២៦] After the implementation of social distancing and stay-at-home orders, many regions have been able to sustain an effective transmission rate ("Rt") of less than one, meaning the disease is in remission in those areas.[១២៧] In a simple model needs on average over time be kept at or below zero to avoid exponential growth.[ត្រូវការអំណះអំណាង]
As a vaccine is not expected until 2021 at the earliest,[១២៨] a key part of managing COVID‑19 is trying to decrease and delay the epidemic peak, known as "flattening the curve".[១១៥] This is done by slowing the infection rate to decrease the risk of health services being overwhelmed, allowing for better treatment of current cases, and delaying additional cases until effective treatments or a vaccine become available.[១១៥][១១៨]
According to the WHO, the use of masks is recommended only if a person is coughing or sneezing or when one is taking care of someone with a suspected infection.[១២៩] For the European Centre for Disease Prevention and Control (ECDC) face masks "... could be considered especially when visiting busy closed spaces ..." but "... only as a complementary measure ..."[១៣០] Several countries have recommended that healthy individuals wear face masks or cloth face coverings (like scarves or bandanas) at least in certain public settings, including China,[១៣១] Hong Kong,[១៣២] Spain,[១៣៣] Italy (Lombardy region),[១៣៤] Russia,[១៣៥] and the United States.[១២២]
Those diagnosed with COVID‑19 or who believe they may be infected are advised by the CDC to stay home except to get medical care, call ahead before visiting a healthcare provider, wear a face mask before entering the healthcare provider's office and when in any room or vehicle with another person, cover coughs and sneezes with a tissue, regularly wash hands with soap and water and avoid sharing personal household items.[២៦][១៣៦] The CDC also recommends that individuals wash hands often with soap and water for at least 20 seconds, especially after going to the toilet or when hands are visibly dirty, before eating and after blowing one's nose, coughing or sneezing. It further recommends using an alcohol-based hand sanitiser with at least 60% alcohol, but only when soap and water are not readily available.[១១៩]
For areas where commercial hand sanitisers are not readily available, the WHO provides two formulations for local production. In these formulations, the antimicrobial activity arises from ethanol or isopropanol. Hydrogen peroxide is used to help eliminate bacterial spores in the alcohol; it is "not an active substance for hand antisepsis". Glycerol is added as a humectant.[១៣៧]
-
Handwashing instructions
-
Prevention efforts are multiplicative, with effects far beyond that of a single spread. Each avoided case leads to more avoided cases down the line, which in turn can stop the outbreak in its tracks.
ការគ្រប់គ្រង
[កែប្រែ]People are managed with supportive care, which may include fluid therapy, oxygen support, and supporting other affected vital organs.[១៣៨][១៣៩][១៤០] The CDC recommends that those who suspect they carry the virus wear a simple face mask.[២៦] Extracorporeal membrane oxygenation (ECMO) has been used to address the issue of respiratory failure, but its benefits are still under consideration.[៣៦][១៤១] Personal hygiene and a healthy lifestyle and diet have been recommended to improve immunity.[១៤២] Supportive treatments may be useful in those with mild symptoms at the early stage of infection.[១៤៣]
The WHO, the Chinese National Health Commission, and the United States' National Institutes of Health have published recommendations for taking care of people who are hospitalised with COVID‑19.[១២៣][១៤៤][១៤៥] Intensivists and pulmonologists in the U.S. have compiled treatment recommendations from various agencies into a free resource, the IBCC.[១៤៦][១៤៧]
Medications
[កែប្រែ]Per the World Health Organization, as of April 2020, there is no specific treatment for COVID‑19.[៥] On May 1, 2020, the United States gave Emergency Use Authorization (not full approval) for remdesivir in people hospitalized with severe COVID-19 after a study suggested it reduced the duration of recovery.[៣០][១៤៨] Researchers continue working on more effective treatments and many vaccine candidates are in development or testing phases.
For symptoms, some medical professionals recommend paracetamol (acetaminophen) over ibuprofen for first-line use.[១៤៩][១៥០][១៥១] The WHO and NIH do not oppose the use of non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen for symptoms,[១២៣][១៥២] and the FDA says currently there is no evidence that NSAIDs worsen COVID‑19 symptoms.[១៥៣]
While theoretical concerns have been raised about ACE inhibitors and angiotensin receptor blockers, as of 19 March 2020, these are not sufficient to justify stopping these medications.[១២៣][១៥៤][១៥៥][១៥៦] One study from April 22 found that people with COVID-19 and hypertension had lower all-cause mortality when on these medications.[១៥៧]
Steroids, such as methylprednisolone, are not recommended unless the disease is complicated by acute respiratory distress syndrome.[១៥៨][១៥៩]
The Australasian Society of Clinical Immunology and Allergy recommend that tocilizumab should be considered as an off-label treatment option for those with COVID-19 related acute respiratory distress syndrome. It recommends this because of its known benefit in cytokine storm caused by a specific cancer treatment, and that cytokine storm may be a significant contributor to mortality in severe COVID-19.[១៦០]
Medications to prevent blood clotting have been suggested for treatment,[៨៥] and anticoagulant therapy with low molecular weight heparin appears to be associated with better outcomes in severe COVID‐19 showing signs of coagulopathy (elevated D-dimer).[១៦១]
Protective equipment
[កែប្រែ]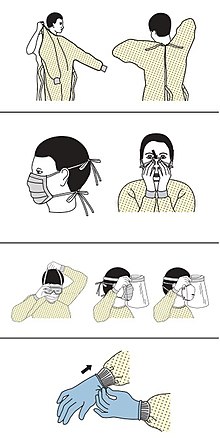
Precautions must be taken to minimise the risk of virus transmission, especially in healthcare settings when performing procedures that can generate aerosols, such as intubation or hand ventilation.[១៦៣] For healthcare professionals caring for people with COVID‑19, the CDC recommends placing the person in an Airborne Infection Isolation Room (AIIR) in addition to using standard precautions, contact precautions, and airborne precautions.[១៦៤]
The CDC outlines the guidelines for the use of personal protective equipment (PPE) during the pandemic. The recommended gear is a PPE gown, respirator or facemask, eye protection, and medical gloves.[១៦៥][១៦៦]
When available, respirators (instead of facemasks) are preferred.[១៦៧] N95 respirators are approved for industrial settings but the FDA has authorised the masks for use under an Emergency Use Authorisation (EUA). They are designed to protect from airborne particles like dust but effectiveness against a specific biological agent is not guaranteed for off-label uses.[១៦៨] When masks are not available, the CDC recommends using face shields or, as a last resort, homemade masks.[១៦៩]
Mechanical ventilation
[កែប្រែ]Most cases of COVID‑19 are not severe enough to require mechanical ventilation or alternatives, but a percentage of cases are.[១៧០][១៧១] The type of respiratory support for individuals with COVID‑19 related respiratory failure is being actively studied for people in the hospital, with some evidence that intubation can be avoided with a high flow nasal cannula or bi-level positive airway pressure.[១៧២] Whether either of these two leads to the same benefit for people who are critically ill is not known.[១៧៣] Some doctors prefer staying with invasive mechanical ventilation when available because this technique limits the spread of aerosol particles compared to a high flow nasal cannula.[១៧០]
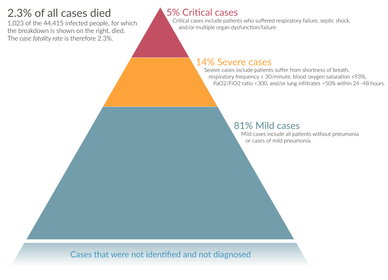
Severe cases are most common in older adults (those older than 60 years,[១៧០] and especially those older than 80 years).[១៧៤] Many developed countries do not have enough hospital beds per capita, which limits a health system's capacity to handle a sudden spike in the number of COVID‑19 cases severe enough to require hospitalisation.[១៧៥] This limited capacity is a significant driver behind calls to flatten the curve.[១៧៥] One study in China found 5% were admitted to intensive care units, 2.3% needed mechanical support of ventilation, and 1.4% died.[៣៦] In China, approximately 30% of people in hospital with COVID‑19 are eventually admitted to ICU.[៣]
Acute respiratory distress syndrome
[កែប្រែ]Mechanical ventilation becomes more complex as acute respiratory distress syndrome (ARDS) develops in COVID‑19 and oxygenation becomes increasingly difficult.[១៧៦] Ventilators capable of pressure control modes and high PEEP[១៧៧] are needed to maximise oxygen delivery while minimising the risk of ventilator-associated lung injury and pneumothorax.[១៧៨] High PEEP may not be available on older ventilators.[ត្រូវការអំណះអំណាង]
| Therapy | Recommendations |
|---|---|
| High-flow nasal oxygen | For SpO2 <93%. May prevent the need for intubation and ventilation |
| Tidal volume | 6mL per kg and can be reduced to 4mL/kg |
| Plateau airway pressure | Keep below 30 cmH2O if possible (high respiratory rate (35 per minute) may be required) |
| Positive end-expiratory pressure | Moderate to high levels |
| Prone positioning | For worsening oxygenation |
| Fluid management | Goal is a negative balance of 0.5–1.0L per day |
| Antibiotics | For secondary bacterial infections |
| Glucocorticoids | Not recommended |
Experimental treatment
[កែប្រែ]Research into potential treatments started in January 2020,[១៧៩] and several antiviral drugs are in clinical trials.[១៨០][១៨១] Remdesivir appears to be the most promising.[១២៦] Although new medications may take until 2021 to develop,[១៨២] several of the medications being tested are already approved for other uses or are already in advanced testing.[១៨៣] Antiviral medication may be tried in people with severe disease.[១៣៨] The WHO recommended volunteers take part in trials of the effectiveness and safety of potential treatments.[១៨៤]
The FDA has granted temporary authorisation to convalescent plasma as an experimental treatment in cases where the person's life is seriously or immediately threatened. It has not undergone the clinical studies needed to show it is safe and effective for the disease.[១៨៥][១៨៦][១៨៧]
បច្ចេកវិទ្យាព័ត៌មាន
[កែប្រែ]In February 2020, China launched a mobile app to deal with the disease outbreak.[១៨៨] Users are asked to enter their name and ID number. The app can detect 'close contact' using surveillance data and therefore a potential risk of infection. Every user can also check the status of three other users. If a potential risk is detected, the app not only recommends self-quarantine, it also alerts local health officials.[១៨៩]
Big data analytics on cellphone data, facial recognition technology, mobile phone tracking, and artificial intelligence are used to track infected people and people whom they contacted in South Korea, Taiwan, and Singapore.[១៩០][១៩១] In March 2020, the Israeli government enabled security agencies to track mobile phone data of people supposed to have coronavirus. The measure was taken to enforce quarantine and protect those who may come into contact with infected citizens.[១៩២] Also in March 2020, Deutsche Telekom shared aggregated phone location data with the German federal government agency, Robert Koch Institute, to research and prevent the spread of the virus.[១៩៣] Russia deployed facial recognition technology to detect quarantine breakers.[១៩៤] Italian regional health commissioner Giulio Gallera said he has been informed by mobile phone operators that "40% of people are continuing to move around anyway".[១៩៥] German government conducted a 48 hours weekend hackathon with more than 42.000 participants.[១៩៦][១៩៧] Two million people in the UK used an app developed in March 2020 by King's College London and Zoe to track people with COVID‑19 symptoms.[១៩៨] Also, the president of Estonia, Kersti Kaljulaid, made a global call for creative solutions against the spread of coronavirus.[១៩៩]
Psychological support
[កែប្រែ]Individuals may experience distress from quarantine, travel restrictions, side effects of treatment, or fear of the infection itself. To address these concerns, the National Health Commission of China published a national guideline for psychological crisis intervention on 27 January 2020.[២០០][២០១]
The Lancet published a 14-page call for action focusing on the UK and stated conditions were such that a range of mental health issues was likely to become more common. BBC quoted Rory O'Connor in saying, "Increased social isolation, loneliness, health anxiety, stress, and an economic downturn are a perfect storm to harm people's mental health and wellbeing."[២០២][២០៣]
Prognosis
[កែប្រែ]| This article relies on references to primary sources or sources affiliated with the subject, rather than references from independent authors and third-party publications. Please add more appropriate citations from reliable sources. (March 2020) |
The severity of COVID‑19 varies. The disease may take a mild course with few or no symptoms, resembling other common upper respiratory diseases such as the common cold. Mild cases typically recover within two weeks, while those with severe or critical diseases may take three to six weeks to recover. Among those who have died, the time from symptom onset to death has ranged from two to eight weeks.[៤១]
Children make up a small proportion of reported cases, with about 1% of cases being under 10 years and 4% aged 10–19 years.[១៨] They are likely to have milder symptoms and a lower chance of severe disease than adults; in those younger than 50 years the risk of death is less than 0.5%, while in those older than 70 it is more than 8%.[២១០][២១១][២១២] Pregnant women may be at higher risk for severe infection with COVID-19 based on data from other similar viruses, like SARS and MERS, but data for COVID-19 is lacking.[២១៣][២១៤] In China, children acquired infections mainly through close contact with their parents or other family members who lived in Wuhan or had traveled there.[២១០]
Some studies have found that the neutrophil to lymphocyte ratio (NLR) may be helpful in early screening for severe illness.[២១៥]
Most of those who die of COVID‑19 have pre-existing (underlying) conditions, including hypertension, diabetes mellitus, and cardiovascular disease.[២១៦] The Istituto Superiore di Sanità reported that out of 8.8% of deaths where medical charts were available for review, 97.2% of sampled patients had at least one comorbidity with the average patient having 2.7 diseases.[២១៧] According to the same report, the median time between the onset of symptoms and death was ten days, with five being spent hospitalised. However, patients transferred to an ICU had a median time of seven days between hospitalisation and death.[២១៧] In a study of early cases, the median time from exhibiting initial symptoms to death was 14 days, with a full range of six to 41 days.[២១៨] In a study by the National Health Commission (NHC) of China, men had a death rate of 2.8% while women had a death rate of 1.7%.[២១៩] Histopathological examinations of post-mortem lung samples show diffuse alveolar damage with cellular fibromyxoid exudates in both lungs. Viral cytopathic changes were observed in the pneumocytes. The lung picture resembled acute respiratory distress syndrome (ARDS).[៤១] In 11.8% of the deaths reported by the National Health Commission of China, heart damage was noted by elevated levels of troponin or cardiac arrest.[៤២] According to March data from the United States, 89% of those hospitalised had preexisting conditions.[២២០]
The availability of medical resources and the socioeconomics of a region may also affect mortality.[២២១] Estimates of the mortality from the condition vary because of those regional differences,[២២២] but also because of methodological difficulties. The under-counting of mild cases can cause the mortality rate to be overestimated.[២២៣] However, the fact that deaths are the result of cases contracted in the past can mean the current mortality rate is underestimated.[២២៤][២២៥] Smokers were 1.4 times more likely to have severe symptoms of COVID‑19 and approximately 2.4 times more likely to require intensive care or die compared to non-smokers.[២២៦]
Concerns have been raised about long-term sequelae of the disease. The Hong Kong Hospital Authority found a drop of 20% to 30% in lung capacity in some people who recovered from the disease, and lung scans suggested organ damage.[២២៧] This may also lead to post-intensive care syndrome following recovery.[២២៨]
| Age | 0–19 | 20–44 | 45–54 | 55–64 | 65–74 | 75–84 | 85+ |
|---|---|---|---|---|---|---|---|
| United States as of 16 March[២៤៤] | 0.0 | 0.1–0.2 | 0.5–0.8 | 1.4–2.6 | 2.7–4.9 | 4.3–10.5 | 10.4–27.3 |
| Note: The lower bound includes all cases. The upper bound excludes cases that were missing data. | |||||||
| 0–9 | 10–19 | 20–29 | 30–39 | 40–49 | 50–59 | 60–69 | 70–79 | 80+ | |
|---|---|---|---|---|---|---|---|---|---|
| Severe disease | ទំព័រគំរូ:Shade (0.0–0.0) |
ទំព័រគំរូ:Shade (0.02–0.08) |
ទំព័រគំរូ:Shade (0.62–2.1) |
ទំព័រគំរូ:Shade (2.0–7.0) |
ទំព័រគំរូ:Shade (2.5–8.7) |
ទំព័រគំរូ:Shade (4.9–17) |
ទំព័រគំរូ:Shade (7.0–24) |
ទំព័រគំរូ:Shade (9.9–34) |
ទំព័រគំរូ:Shade (11–38) |
| Death | ទំព័រគំរូ:Shade (0.00016–0.025) |
ទំព័រគំរូ:Shade (0.0015–0.050) |
ទំព័រគំរូ:Shade (0.014–0.092) |
ទំព័រគំរូ:Shade (0.041–0.19) |
ទំព័រគំរូ:Shade (0.076–0.32) |
ទំព័រគំរូ:Shade (0.34–1.3) |
ទំព័រគំរូ:Shade (1.1–3.9) |
ទំព័រគំរូ:Shade (2.5–8.4) |
ទំព័រគំរូ:Shade (3.8–13) |
| Total infection fatality rate is estimated to be 0.66% (0.39–1.3). Infection fatality rate is fatality per all infected individuals, regardless of whether they were diagnosed or had any symptoms. Numbers in parentheses are 95% credible intervals for the estimates. | |||||||||
ភាពស៊ាំ
[កែប្រែ]It is unknown (as of April 2020) if past infection provides effective and long-term immunity in people who recover from the disease.[២៤៦][២៤៧] Some of the infected have been reported to develop protective antibodies, so acquired immunity is presumed likely, based on the behaviour of other coronaviruses.[២៤៨] However, cases in which recovery from COVID‑19 was followed by positive tests for coronavirus at a later date have been reported.[២៤៩][២៥០][២៥១][២៥២] These cases are believed to be lingering infection rather than reinfection,[២៥២] or false positives due to remaining RNA fragments.[២៥៣] Some other coronaviruses circulating in people are capable of reinfection after roughly a year.[២៥៤][២៥៥]
ប្រវត្តិ
[កែប្រែ]The virus is thought to be natural and has an animal origin,[៧១] through spillover infection.[២៥៦] The actual origin is unknown, but the first known cases of infection happened in China. By December 2019, the spread of infection was almost entirely driven by human-to-human transmission.[២០៥][២៥៧] A study of the first 41 cases of confirmed COVID‑19, published in January 2020 in The Lancet, revealed the earliest date of onset of symptoms as 1 December 2019.[២៥៨][២៥៩][២៦០] Official publications from the WHO reported the earliest onset of symptoms as 8 December 2019.[២៦១] Human-to-human transmission was confirmed by the WHO and Chinese authorities by 20 January 2020.[២៦២][២៦៣]
រោគរាតត្បាតវិទ្យា
[កែប្រែ]Several measures are commonly used to quantify mortality.[២៦៤] These numbers vary by region and over time and are influenced by the volume of testing, healthcare system quality, treatment options, time since the initial outbreak, and population characteristics such as age, sex, and overall health.[២៦៥]
The death-to-case ratio reflects the number of deaths divided by the number of diagnosed cases within a given time interval. Based on Johns Hopkins University statistics, the global death-to-case ratio is ២% (២២៦៥៥៥៩/១០៤៣៣៣៨៧៨) as of ៤ កុម្ភៈ ២០២១.[៧] The number varies by region.[២៦៦]
Other measures include the case fatality rate (CFR), which reflects the percent of diagnosed individuals who die from a disease, and the infection fatality rate (IFR), which reflects the percent of infected individuals (diagnosed and undiagnosed) who die from a disease. These statistics are not time-bound and follow a specific population from infection through case resolution. Many academics have attempted to calculate these numbers for specific populations.[២៦៧]
Outbreaks have occurred in prisons due to crowding and an inability to enforce adequate social distancing.[២៦៨] In the United States, the prisoner population is aging and many of them are at high risk for poor outcomes from COVID-19 due to high rates of coexisting heart and lung disease, and poor access to high-quality healthcare.[២៦៨]
Infection fatality rate
[កែប្រែ]Our World in Data states that as of March 25, 2020, the infection fatality rate (IFR) cannot be accurately calculated.[២៧១] In February, the World Health Organization estimated the IFR at 0.94%, with a confidence interval between 0.37 percent to 2.9 percent.[២៧២] The University of Oxford Centre for Evidence-Based Medicine (CEBM) estimated a global CFR of 0.72 percent and IFR of 0.1 percent to 0.36 percent.[២៧៣] According to CEBM, random antibody testing in Germany suggested an IFR of 0.37% (0.12% to 0.87%) there, but there have been concerns about false positives.[២៧៣][២៧៤][២៧៥] Firm lower limits of infection fatality rates have been established in a number of locations. In New York City, with a population of 8.4 million, as of April 25, 16,673 (0.20% of the population) have died from COVID-19, and 20,800 (0.25% of the population) excess deaths have occurred, and in Bergamo province, where 0.57% of the population has died.[២៧៦][២៧៧][២៧៨] To get a better view on the number of people infected initial antibody testing have been carried out, but there are no valid scientific reports based on any of them as of yet.[២៧៩][២៨០]
ភាពខុសគ្នាប្រាំមួយយ៉ាង
[កែប្រែ]The impact of the pandemic and its mortality rate are different for men and women.[២៨១] Mortality is higher in men in studies conducted in China and Italy.[១][២៨២][២៨៣] The higher risk for men appears in their 50s, and begins to taper off only at 90.[២៨៣] In China, the death rate was 2.8 percent for men and 1.7 percent for women.[២៨៣] The exact reasons for this sex-difference are not known, but genetic and behavioural factors could be a reason.[២៨១] Sex-based immunological differences, a lower prevalence of smoking in women, and men developing co-morbid conditions such as hypertension at a younger age than women could have contributed to the higher mortality in men.[២៨៣] In Europe, of those infected with COVID‑19, 57% were men; of those infected with COVID‑19 who also died, 72% were men.[២៨៤] As of April 2020, the U.S. government is not tracking sex-related data of COVID‑19 infections.[២៨៥] Research has shown that viral illnesses like Ebola, HIV, influenza, and SARS affect men and women differently.[២៨៥] A higher percentage of health workers, particularly nurses, are women, and they have a higher chance of being exposed to the virus.[២៨៦] School closures, lockdowns, and reduced access to healthcare following the 2019–20 coronavirus pandemic may differentially affect the genders and possibly exaggerate existing gender disparity.[២៨១][២៨៧]
Ethnic differences
[កែប្រែ]In the U.S., a greater proportion of deaths due to COVID-19 have occurred among African Americans.[២៨៨] Structural factors that prevent African Americans from practicing social distancing include their concentration in crowded substandard housing and "essential" occupations such as public transit employees and health-care workers. Greater prevalence of lacking health insurance and care and of underlying conditions such as diabetes, hypertension and heart disease also increase their risk of death.[២៨៩] Similar issues affect Native American and Latino communities.[២៨៨] Leaders have called for efforts to research and address the disparities.[២៩០]
Existing respiratory problems
[កែប្រែ]When someone with existing respiratory problems is infected with COVID-19, they are at greater risk for severe symptoms.[២៩១] COVID-19 also poses a greater risk to people who misuse opioids and methamphetamines, insofar as their drug use may have caused lung damage.[២៩២]
សង្គម និង វប្បធម៌
[កែប្រែ]ឈ្មោះ
[កែប្រែ]During the initial outbreak in Wuhan, China, the virus and disease were commonly referred to as "coronavirus" and "Wuhan coronavirus",[២៩៣][២៩៤][២៩៥] with the disease sometimes called "Wuhan pneumonia".[២៩៦][២៩៧] In the past, many diseases have been named after geographical locations, such as the Spanish flu,[២៩៨] Middle East Respiratory Syndrome, and Zika virus.[២៩៩]
In January 2020, the World Health Organisation recommended 2019-nCov[៣០០] and 2019-nCoV acute respiratory disease[៣០១] as interim names for the virus and disease per 2015 guidance and international guidelines against using geographical locations (e.g. Wuhan, China), animal species or groups of people in disease and virus names to prevent social stigma.[៣០២][៣០៣][៣០៤]
The official names COVID‑19 and SARS-CoV-2 were issued by the WHO on 11 February 2020.[៣០៥] WHO chief Tedros Adhanom Ghebreyesus explained: CO for corona, VI for virus, D for disease and 19 for when the outbreak was first identified (31 December 2019).[៣០៦] The WHO additionally uses "the COVID‑19 virus" and "the virus responsible for COVID‑19" in public communications.[៣០៥] Both the disease and virus are commonly referred to as "coronavirus" in the media and public discourse.
Misinformation
[កែប្រែ]After the initial outbreak of COVID‑19, conspiracy theories, misinformation and disinformation emerged regarding the origin, scale, prevention, treatment and other aspects of the disease and rapidly spread online.[៣០៧][៣០៨][៣០៩]
Decreased emergency room use
[កែប្រែ]In Austria, 39% fewer persons sought help for cardiac symptoms in the month of March. A study estimated that there were 110 incidents of preventable cardiac death as compared to 86 confirmed deaths from Coronavirus as of March 29.[៣១០]
A preliminary study in the U.S. found 38% under-utilization of cardiac care units as compared to normal.[៣១១] The head of cardiology at the University of Arizona has stated, "My worry is some of these people are dying at home because they're too scared to go to the hospital."[៣១២] There is also concern that persons with symptoms of stroke and appendicitis are delaying seeking help.[៣១២][៣១៣]
សត្វផ្សេងៗ
[កែប្រែ]Humans appear to be capable of spreading the virus to some other animals. A domestic cat in Liège, Belgium, tested positive after it started showing symptoms (diarrhoea, vomiting, shortness of breath) a week later than its owner, who was also positive.[៣១៤] Tigers at the Bronx Zoo in New York, United States, tested positive for the virus and showed symptoms of COVID‑19, including a dry cough and loss of appetite.[៣១៥] Minks at two farms in the Netherlands also tested positive for COVID-19.[៣១៦]
A study on domesticated animals inoculated with the virus found that cats and ferrets appear to be "highly susceptible" to the disease, while dogs appear to be less susceptible, with lower levels of viral replication. The study failed to find evidence of viral replication in pigs, ducks, and chickens.[៣១៧]
ការស្រាវជ្រាវ
[កែប្រែ]No medication or vaccine is approved to treat the disease.[១៨៣] International research on vaccines and medicines in COVID‑19 is underway by government organisations, academic groups, and industry researchers.[៣១៨][៣១៩] In March, the World Health Organisation initiated the "SOLIDARITY Trial" to assess the treatment effects of four existing antiviral compounds with the most promise of efficacy.[៣២០]
There has been a great deal of COVID-19 research, involving accelerated research processes and publishing shortcuts to meet the global demand. To minimise the impact of misinformation, medical professionals and the public are advised to expect rapid changes to available information, and to be attentive to retractions and other updates.[៣២១]
វ៉ាក់សាំង
[កែប្រែ]There is no available vaccine, but various agencies are actively developing vaccine candidates. Previous work on SARS-CoV is being used because both SARS-CoV and SARS-CoV-2 use the ACE2 receptor to enter human cells.[៣២២] Three vaccination strategies are being investigated. First, researchers aim to build a whole virus vaccine. The use of such a virus, be it inactive or dead, aims to elicit a prompt immune response of the human body to a new infection with COVID‑19. A second strategy, subunit vaccines, aims to create a vaccine that sensitises the immune system to certain subunits of the virus. In the case of SARS-CoV-2, such research focuses on the S-spike protein that helps the virus intrude the ACE2 enzyme receptor. A third strategy is that of the nucleic acid vaccines (DNA or RNA vaccines, a novel technique for creating a vaccination). Experimental vaccines from any of these strategies would have to be tested for safety and efficacy.[៣២៣]
On 16 March 2020, the first clinical trial of a vaccine started with four volunteers in Seattle, United States. The vaccine contains a harmless genetic code copied from the virus that causes the disease.[៣២៤]
Antibody-dependent enhancement has been suggested as a potential challenge for vaccine development for SARS-COV-2, but this is controversial.[៣២៥]
Medications
[កែប្រែ]At least 29 phase II–IV efficacy trials in COVID‑19 were concluded in March 2020 or scheduled to provide results in April from hospitals in China.[៣២៦][៣២៧] There are more than 300 active clinical trials underway as of April 2020.[១២៦] Seven trials were evaluating already approved treatments, including four studies on hydroxychloroquine or chloroquine.[៣២៧] Repurposed antiviral drugs make up most of the Chinese research, with nine phase III trials on remdesivir across several countries due to report by the end of April.[៣២៦][៣២៧] Other candidates in trials include vasodilators, corticosteroids, immune therapies, lipoic acid, bevacizumab, and recombinant angiotensin-converting enzyme 2.[៣២៧]
The COVID‑19 Clinical Research Coalition has goals to 1) facilitate rapid reviews of clinical trial proposals by ethics committees and national regulatory agencies, 2) fast-track approvals for the candidate therapeutic compounds, 3) ensure standardised and rapid analysis of emerging efficacy and safety data and 4) facilitate sharing of clinical trial outcomes before publication.[៣២៨][៣២៩]
Several existing medications are being evaluated for the treatment of COVID‑19,[១៨៣] including remdesivir, chloroquine, hydroxychloroquine, lopinavir/ritonavir, and lopinavir/ritonavir combined with interferon beta.[៣២០][៣៣០] There is tentative evidence for efficacy by remdesivir, as of March 2020.[៣៣១][៣៣២] Clinical improvement was observed in patients treated with compassionate-use remdesivir.[៣៣៣] Remdesivir inhibits SARS-CoV-2 in vitro.[៣៣៤] Phase III clinical trials are underway in the U.S., China, and Italy.[១៨៣][៣២៦][៣៣៥]
In 2020, a trial found that lopinavir/ritonavir was ineffective in the treatment of severe illness.[៣៣៦] Nitazoxanide has been recommended for further in vivo study after demonstrating low concentration inhibition of SARS-CoV-2.[៣៣៤]
There are mixed results as of 3 April 2020 as to the effectiveness of hydroxychloroquine as a treatment for COVID‑19, with some studies showing little or no improvement.[៣៣៧][៣៣៨] The studies of chloroquine and hydroxychloroquine with or without azithromycin have major limitations that have prevented the medical community from embracing these therapies without further study.[១២៦]
Oseltamivir does not inhibit SARS-CoV-2 in vitro and has no known role in COVID‑19 treatment.[១២៦]
Anti-cytokine storm
[កែប្រែ]Cytokine release syndrome (CRS) can be a complication in the later stages of severe COVID‑19. There is preliminary evidence that hydroxychloroquine may have anti-cytokine storm properties.[៣៣៩]
Tocilizumab has been included in treatment guidelines by China's National Health Commission after a small study was completed.[៣៤០][៣៤១] It is undergoing a phase 2 non-randomised trial at the national level in Italy after showing positive results in people with severe disease.[៣៤២][៣៤៣] Combined with a serum ferritin blood test to identify cytokine storms, it is meant to counter such developments, which are thought to be the cause of death in some affected people.[៣៤៤][៣៤៥][១៣] The interleukin-6 receptor antagonist was approved by the FDA to undergo a phase III clinical trial assessing the medication's impact on COVID‑19 based on retrospective case studies for the treatment of steroid-refractory cytokine release syndrome induced by a different cause, CAR T cell therapy, in 2017.[៣៤៦] To date, there is no randomised, controlled evidence that tocilizumab is an efficacious treatment for CRS. Prophylactic tocilizumab has been shown to increase serum IL-6 levels by saturating the IL-6R, driving IL-6 across the blood-brain barrier, and exacerbating neurotoxicity while having no impact on the incidence of CRS.[៣៤៧]
Lenzilumab, an anti-GM-CSF monoclonal antibody, is protective in murine models for CAR T cell-induced CRS and neurotoxicity and is a viable therapeutic option due to the observed increase of pathogenic GM-CSF secreting T-cells in hospitalised patients with COVID‑19.[៣៤៨]
The Feinstein Institute of Northwell Health announced in March a study on "a human antibody that may prevent the activity" of IL-6.[៣៤៩]
Passive antibodies
[កែប្រែ]Transferring purified and concentrated antibodies produced by the immune systems of those who have recovered from COVID‑19 to people who need them is being investigated as a non-vaccine method of passive immunisation.[៣៥០] This strategy was tried for SARS with inconclusive results.[៣៥០] Viral neutralisation is the anticipated mechanism of action by which passive antibody therapy can mediate defence against SARS-CoV-2. Other mechanisms, however, such as antibody-dependent cellular cytotoxicity and/or phagocytosis, may be possible.[៣៥០] Other forms of passive antibody therapy, for example, using manufactured monoclonal antibodies, are in development.[៣៥០] Production of convalescent serum, which consists of the liquid portion of the blood from recovered patients and contains antibodies specific to this virus, could be increased for quicker deployment.[៣៥១]
សូមមើលផង
[កែប្រែ]- Coronavirus diseases, a group of closely related syndromes
- Coronavirus recession
- Disease X, a WHO term
- Li Wenliang, a doctor at Central Hospital of Wuhan who died of COVID-19 after raising awareness of its spread
- List of unproven methods against COVID-19
កំណត់
[កែប្រែ]ឯកសារយោង
[កែប្រែ]- ↑ ១,០ ១,១ ១,២ (February 2020)"Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study". Lancet 395 (10223): 507–513. DOI:10.1016/S0140-6736(20)30211-7.
- ↑ (March 2020)"Novel Coronavirus Pneumonia (COVID-19) Progression Course in 17 Discharged Patients: Comparison of Clinical and Thin-Section CT Features During Recovery". Clinical Infectious Diseases. DOI:10.1093/cid/ciaa271.
- ↑ ៣,០ ៣,១ ៣,២ ៣,៣ ៣,៤ ៣,៥ "Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19)". Centers for Disease Control and Prevention. 6 ខែមេសា 2020. Archived from the original on 2 ខែមីនា 2020. Retrieved 19 ខែមេសា 2020.
- ↑ ៤,០ ៤,១ ៤,២ ៤,៣ "Symptoms of Coronavirus". U.S. Centers for Disease Control and Prevention (CDC). 20 ខែមីនា 2020. Archived from the original on 30 ខែមករា 2020.
- ↑ ៥,០០ ៥,០១ ៥,០២ ៥,០៣ ៥,០៤ ៥,០៥ ៥,០៦ ៥,០៧ ៥,០៨ ៥,០៩ ៥,១០ ៥,១១ ៥,១២ ៥,១៣ ៥,១៤ ៥,១៥ "Q&A on coronaviruses". World Health Organization. 8 ខែមេសា 2020. Archived from the original on 20 ខែមករា 2020. Retrieved 30 ខែមេសា 2020.
- ↑ ៦,០ ៦,១ (8 April 2020)"Quarantine alone or in combination with other public health measures to control COVID-19: a rapid review.". The Cochrane Database of Systematic Reviews 4: CD013574. DOI:10.1002/14651858.CD013574.
- ↑ ៧,០ ៧,១ ៧,២ ៧,៣ "COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU)". ArcGIS. Johns Hopkins University. Retrieved 23 ខែមេសា 2020.
- ↑ "Coronavirus disease 2019 (COVID-19)—Symptoms and causes". Mayo Clinic. Retrieved 14 ខែមេសា 2020.
- ↑ ៩,០ ៩,១ ៩,២ ៩,៣ (February 2020)"The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health - The latest 2019 novel coronavirus outbreak in Wuhan, China". Int J Infect Dis 91: 264–66. DOI:10.1016/j.ijid.2020.01.009.
- ↑ ១០,០ ១០,១ "WHO Director-General's opening remarks at the media briefing on COVID-19"។ Press release។ 11 March 2020។ https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020។ បានយកមក 12 March 2020។
- ↑ "Q&A on coronaviruses (COVID-19)". World Health Organization (WHO). Archived from the original on 20 ខែមករា 2020. Retrieved 11 ខែមីនា 2020.
- ↑ ១២,០ ១២,១ Hopkins, Claire. "Loss of sense of smell as marker of COVID-19 infection" (PDF). Ear, Nose and Throat surgery body of United Kingdom. Archived from the original on 27 ខែឧសភា 2020. Retrieved 28 ខែមីនា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help) - ↑ ១៣,០ ១៣,១ (March 2020)"COVID-19: consider cytokine storm syndromes and immunosuppression". Lancet 395 (10229): 1033–1034. DOI:10.1016/S0140-6736(20)30628-0.
- ↑ "Q&A on coronaviruses". World Health Organization (WHO). Archived from the original on 20 ខែមករា 2020. Retrieved 27 ខែមករា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (March 2020)"The COVID-19 epidemic". Tropical Medicine & International Health n/a (n/a): 278–80. DOI:10.1111/tmi.13383.
- ↑ ១៦,០ ១៦,១ ១៦,២ ១៦,៣ ១៦,៤ ១៦,៥ "How COVID-19 Spreads". Centers for Disease Control and Prevention (CDC). 2 ខែមេសា 2020. Archived from the original on 3 ខែមេសា 2020. Retrieved 3 ខែមេសា 2020.
- ↑ (March 2020)"Turbulent Gas Clouds and Respiratory Pathogen Emissions: Potential Implications for Reducing Transmission of COVID-19". JAMA. DOI:10.1001/jama.2020.4756.
- ↑ ១៨,០០ ១៨,០១ ១៨,០២ ១៨,០៣ ១៨,០៤ ១៨,០៥ ១៨,០៦ ១៨,០៧ ១៨,០៨ ១៨,០៩ ១៨,១០ ១៨,១១ "Q & A on COVID-19". European Centre for Disease Prevention and Control. Retrieved 30 ខែមេសា 2020.
- ↑ "New coronavirus stable for hours on surfaces". National Institutes of Health. 17 ខែមីនា 2020. Archived from the original on 23 ខែមីនា 2020. Retrieved 30 ខែមេសា 2020.
- ↑ "Coronavirus disease 2019 (COVID-19) Situation Report—73" (PDF). World Health Organization. 2 ខែមេសា 2020. Retrieved 3 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ ២១,០ ២១,១ "Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens from Persons for Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention (CDC). 11 ខែកុម្ភៈ 2020. Archived from the original on 4 ខែមីនា 2020. Retrieved 26 ខែមីនា 2020.
- ↑ ២២,០ ២២,១ ២២,២ ២២,៣ ២២,៤ (March 2020)"Coronavirus Disease 2019 (COVID-19): A Systematic Review of Imaging Findings in 919 Patients". American Journal of Roentgenology: 1–7. DOI:10.2214/AJR.20.23034.
- ↑ ២៣,០ ២៣,១ "ACR Recommendations for the use of Chest Radiography and Computed Tomography (CT) for Suspected COVID-19 Infection". American College of Radiology. 22 ខែមីនា 2020. Archived from the original on 28 ខែមីនា 2020.
- ↑ "Advice for public". World Health Organization (WHO). Archived from the original on 26 ខែមករា 2020. Retrieved 25 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Guidance on social distancing for everyone in the UK". GOV.UK. Archived from the original on 24 ខែមីនា 2020. Retrieved 25 ខែមីនា 2020.
- ↑ ២៦,០ ២៦,១ ២៦,២ Centers for Disease Control and Prevention (5 ខែមេសា 2020). "What to Do if You Are Sick". Centers for Disease Control and Prevention (CDC). Archived from the original on 14 ខែកុម្ភៈ 2020. Retrieved 24 ខែមេសា 2020.
- ↑ ២៧,០ ២៧,១ "When and how to use masks". World Health Organization (WHO). Archived from the original on 7 ខែមីនា 2020. Retrieved 24 ខែមេសា 2020.
- ↑ (March 2020)"Rational use of face masks in the COVID-19 pandemic". Lancet Respiratory Medicine 0 (5): 434–436. DOI:10.1016/S2213-2600(20)30134-X.
- ↑ Tait, Robert (2020-03-30). "Czechs get to work making masks after government decree". The Guardian. ល.ត.ម.អ. 0261-3077. https://www.theguardian.com/world/2020/mar/30/czechs-get-to-work-making-masks-after-government-decree-coronavirus.
- ↑ ៣០,០ ៣០,១ "Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment"។ Press release។ 1 May 2020។ https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-issues-emergency-use-authorization-potential-covid-19-treatment។ បានយកមក 1 May 2020។
- ↑ "How to Protect Yourself & Others". Centers for Disease Control and Prevention (CDC). 8 ខែមេសា 2020. Archived from the original on 26 ខែកុម្ភៈ 2020. Retrieved 9 ខែមេសា 2020.
- ↑ "Statement on the second meeting of the International Health Regulations (2005) Emergency Committee regarding the outbreak of novel coronavirus (2019-nCoV)". World Health Organization (WHO). Archived from the original on 31 ខែមករា 2020. Retrieved 11 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Hundreds of evacuees to be held on bases in California; Hong Kong and Taiwan restrict travel from mainland China". The Washington Post. 6 February 2020. https://www.washingtonpost.com/world/asia_pacific/coronavirus-china-live-updates/2020/02/05/114ced8a-479c-11ea-bc78-8a18f7afcee7_story.html.
- ↑ "WHO Situation Report #87" (PDF). WHO. 16 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ (24 February 2020)"Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study". Lancet 8 (5): 475–481. DOI:10.1016/S2213-2600(20)30079-5.
- ↑ ៣៦,០ ៣៦,១ ៣៦,២ (February 2020)"Clinical Characteristics of Coronavirus Disease 2019 in China". The New England Journal of Medicine 382 (18): 1708–1720. DOI:10.1056/nejmoa2002032.
- ↑ Hessen, Margaret Trexler (27 ខែមករា 2020). "Novel Coronavirus Information Center: Expert guidance and commentary". Elsevier Connect. Archived from the original on 30 ខែមករា 2020. Retrieved 31 ខែមករា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Wei, Xiao-Shan; Wang, Xuan; Niu, Yi-Ran; Ye, Lin-Lin; Peng, Wen-Bei; Wang, Zi-Hao; Yang, Wei-Bing; Yang, Bo-Han 7et al (26 February 2020). "Clinical Characteristics of SARS-CoV-2 Infected Pneumonia with Diarrhea". អ.វ.ល.:10.2139/ssrn.3546120.
- ↑ ៣៩,០ ៣៩,១ ៣៩,២ (February 2020)"Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China". Lancet 395 (10223): 497–506. DOI:10.1016/S0140-6736(20)30183-5.
- ↑ (1 March 2020)"Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges". International Journal of Antimicrobial Agents 55 (3). ISSN 0924-8579. DOI:10.1016/j.ijantimicag.2020.105924.
- ↑ ៤១,០ ៤១,១ ៤១,២ ៤១,៣ Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19) (Report). World Health Organization (WHO). 16–24 February 2020. https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf។ បានយកមក 21 March 2020.
- ↑ ៤២,០ ៤២,១ (March 2020)"COVID-19 and the cardiovascular system". Nature Reviews. Cardiology 17 (5): 259–260. DOI:10.1038/s41569-020-0360-5.
- ↑ (April 15, 2020)"Smell and taste dysfunction in patients with COVID-19". Lancet Infectious Diseases. DOI:10.1016/S1473-3099(20)30293-0. Retrieved on 21 April 2020.
- ↑ "Symptoms of Coronavirus". Centers for Disease Control and Prevention. 27 ខែមេសា 2020. Retrieved 28 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ (2020). "Sixty seconds on ... anosmia". BMJ 368: m1202. ISSN 1756-1833. DOI:10.1136/bmj.m1202.
- ↑ (19 February 2020)"Coronavirus disease 2019 (COVID-19): situation report, 29".
- ↑ "Q&A on coronaviruses (COVID-19): How long is the incubation period for COVID-19?". World Health Organization (WHO). Archived from the original on 20 ខែមករា 2020. Retrieved 26 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (10 March 2020)"The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application". Annals of Internal Medicine 172 (9). ISSN 0003-4819. DOI:10.7326/M20-0504.
- ↑ (March 16, 2020)"Report 9: Impact of non-pharmaceutical interventions (NPIs) to reduce COVID-19 mortality and healthcare demand". Retrieved on April 21, 2020.
- ↑ (2020)"Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020". Euro Surveillance 25 (10). DOI:10.2807/1560-7917.ES.2020.25.10.2000180. Retrieved on April 21, 2020.
- ↑ "Clinical Questions about COVID-19: Questions and Answers". Centers for Disease Control and Prevention. 11 ខែកុម្ភៈ 2020. Archived from the original on 14 ខែកុម្ភៈ 2020. Retrieved 31 ខែមីនា 2020.
- ↑ (March 4, 2020)"Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): Facts and myths". Journal of Microbiology, Immunology, and Infection. DOI:10.1016/j.jmii.2020.02.012.
- ↑ Bai, Yan (2020-02-21). "Presumed Asymptomatic Carrier Transmission of COVID-19". JAMA 323 (14). ISSN 0098-7484. DOI:10.1001/jama.2020.2565.
- ↑ ៥៤,០ ៥៤,១ "China Reveals 1,541 Symptom-Free Virus Cases Under Pressure". www.bloomberg.com. 31 ខែមីនា 2020. Retrieved 31 ខែមីនា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ "코로나19 국내 발생현황 브리핑 (20. 03. 16. 14시)". ktv.go.kr (in កូរ៉េ). Retrieved 31 ខែមីនា 2020.
- ↑ (1 May 2020)"Neurological complications of coronavirus and COVID-19.". Revista de neurologia 70 (9): 311–322. DOI:10.33588/rn.7009.2020179.
- ↑ (18 April 2020)"Cardiovascular complications in COVID-19.". The American Journal of Emergency Medicine. DOI:10.1016/j.ajem.2020.04.048.
- ↑ (February 2020)"COVID-19: what is next for public health?". Lancet 395 (10224): 542–545. DOI:10.1016/s0140-6736(20)30374-3.
- ↑ "Features, Evaluation and Treatment Coronavirus (COVID-19)". StatPearls. Treasure Island (FL): StatPearls Publishing. 2020. https://www.ncbi.nlm.nih.gov/books/NBK554776/។ បានយកមក 18 March 2020.
- ↑ (2020)"Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study". The Lancet 395 (10229): 1054–1062. ISSN 0140-6736. DOI:10.1016/s0140-6736(20)30566-3.
- ↑ (April 13, 2020)"Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19): A Review". JAMA. DOI:10.1001/jama.2020.6019.
- ↑ (March 2020)"Liver injury during highly pathogenic human coronavirus infections". Liver International 40 (5): 998–1004. DOI:10.1111/liv.14435.
- ↑ (March 2020)"Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1". New England Journal of Medicine 382 (16): 1564–7. ISSN 0028-4793. DOI:10.1056/NEJMc2004973.
- ↑ CDC (24 ខែមេសា 2020). "Household cleaners and disinfectants can cause health problems when not used properly. Follow the instructions on the product label to ensure safe and effective use. Learn more about cleaning and disinfecting your home: https://bit.ly/2S2a9yc .pic.twitter.com/rdcR9QJNIR". @cdcgov (in អង់គ្លេស). Retrieved 6 ខែឧសភា 2020.
- ↑ ៦៥,០ ៦៥,១ (12 February 2020)"Consistent Detection of 2019 Novel Coronavirus in Saliva". Clinical Infectious Diseases. DOI:10.1093/cid/ciaa149.
- ↑ "Coronavirus disease 2019 (COVID-19) Situation Report—73" (PDF). World Health Organization. 2 ខែមេសា 2020. Retrieved 3 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ Coronavirus disease 2019 (COVID-19) in the EU/EEA and the UK – ninth update, 23 April 2020. Stockholm: ECDC, p.8-9.
- ↑ "COVID-19 and Our Communities -ACON – We are a New South Wales based health promotion organisation specialising in HIV prevention, HIV support and lesbian, gay, bisexual, transgender and intersex (LGBTI) health". Acon.org.au. Retrieved 29 ខែមេសា 2020.
- ↑ "Sex and Coronavirus Disease 2019 (COVID-19)" (PDF). nyc.gov. 27 ខែមីនា 2020. Retrieved 29 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ "Outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): increased transmission beyond China—fourth update" (PDF). European Centre for Disease Prevention and Control. 14 ខែកុម្ភៈ 2020. Retrieved 8 ខែមីនា 2020.
- ↑ ៧១,០ ៧១,១ (17 March 2020)"The proximal origin of SARS-CoV-2". Nature Medicine 26 (4): 450–452. ISSN 1546-170X. DOI:10.1038/s41591-020-0820-9.
- ↑ (February 2020)"A Novel Coronavirus from Patients with Pneumonia in China, 2019". The New England Journal of Medicine 382 (8): 727–733. DOI:10.1056/NEJMoa2001017.
- ↑ (26 February 2020)"Mystery deepens over animal source of coronavirus". Nature 579 (7797): 18–19. DOI:10.1038/d41586-020-00548-w.
- ↑ (2020)"Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses". Nature Microbiology 5 (4): 562–569. DOI:10.1038/s41564-020-0688-y.
- ↑ (March 2020)"Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target". Intensive Care Medicine 46 (4): 586–590. DOI:10.1007/s00134-020-05985-9.
- ↑ ៧៦,០ ៧៦,១ (February 2020)"High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa". International Journal of Oral Science 12 (1). DOI:10.1038/s41368-020-0074-x.
- ↑ (March 2020)"Angiotensin receptor blockers as tentative SARS‐CoV‐2 therapeutics". Drug Development Research. DOI:10.1002/ddr.21656.
- ↑ Li, Yan-Chao (2020). "The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients". Journal of Medical Virology 92 (6): 552–555. ISSN 1096-9071. DOI:10.1002/jmv.25728.
- ↑ Baig, Abdul Mannan (March 2020). "Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host–Virus Interaction, and Proposed Neurotropic Mechanisms". ACS Chemical Neuroscience 11 (7): 995–998. ISSN 1948-7193. DOI:10.1021/acschemneuro.0c00122.
- ↑ (27 February 2020)"COVID-19: Gastrointestinal manifestations and potential fecal-oral transmission". Gastroenterology 158 (6): 1518–1519. ISSN 0016-5085. DOI:10.1053/j.gastro.2020.02.054.
- ↑ (2004)"Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis". The Journal of Pathology 203 (2): 631–637. ISSN 1096-9896. DOI:10.1002/path.1570.
- ↑ ៨២,០ ៨២,១ ៨២,២ Zheng, Ying-Ying (March 2020). "COVID-19 and the cardiovascular system". Nature Reviews Cardiology 17 (5): 259–260. ISSN 1759-5010. DOI:10.1038/s41569-020-0360-5.
- ↑ Wang, Dawei (March 2020). "Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China". JAMA 323 (11): 1061–1069. ISSN 0098-7484. DOI:10.1001/jama.2020.1585.
- ↑ Turner, Anthony J. (2004-06-01). "ACE2: from vasopeptidase to SARS virus receptor". Trends in Pharmacological Sciences 25 (6): 291–294. ISSN 0165-6147. DOI:10.1016/j.tips.2004.04.001.
- ↑ ៨៥,០ ៨៥,១ Klok, F.A. (April 2020). "Incidence of thrombotic complications in critically ill ICU patients with COVID-19". Thrombosis Research. ISSN 0049-3848. DOI:10.1016/j.thromres.2020.04.013.
- ↑ Cui, Songping (2020-04-09). "Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia". Journal of Thrombosis and Haemostasis. DOI:10.1111/jth.14830.
- ↑ ៨៧,០ ៨៧,១ Wadman, Meredith (April 2020). "How does coronavirus kill? Clinicians trace a ferocious rampage through the body, from brain to toes". Science. DOI:10.1126/science.abc3208.
- ↑ (10 April 2020)"Human Kidney is a Target for Novel Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection". MedRxiv: 2020.03.04.20031120. DOI:10.1101/2020.03.04.20031120.
- ↑ ៨៩,០ ៨៩,១ (April 2020)"COVID-19 autopsies, Oklahoma, USA". American Journal of Clinical Pathology 153 (6): 725–733. DOI:10.1093/ajcp/aqaa062.
- ↑ (March 2020)"The cytokine release syndrome (CRS) of severe COVID-19 and Interleukin-6 receptor (IL-6R) antagonist Tocilizumab may be the key to reduce the mortality". International Journal of Antimicrobial Agents. DOI:10.1016/j.ijantimicag.2020.105954.
- ↑ (2020). "Aberrant pathogenic GM-CSF+ T cells and inflammatory CD14+CD16+ monocytes in severe pulmonary syndrome patients of a new coronavirus". bioRxiv Pre-print: 2020.02.12.945576. DOI:10.1101/2020.02.12.945576.
- ↑ "CDC Tests for 2019-nCoV". Centers for Disease Control and Prevention. 5 ខែកុម្ភៈ 2020. Archived from the original on 14 ខែកុម្ភៈ 2020. Retrieved 12 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases". World Health Organization (WHO). Archived from the original on 17 ខែមីនា 2020. Retrieved 13 ខែមីនា 2020.
- ↑ "2019 Novel Coronavirus (2019-nCoV) Situation Summary". Centers for Disease Control and Prevention. 30 ខែមករា 2020. Archived from the original on 26 ខែមករា 2020. Retrieved 30 ខែមករា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Real-Time RT-PCR Panel for Detection 2019-nCoV". Centers for Disease Control and Prevention. 29 ខែមករា 2020. Archived from the original on 30 ខែមករា 2020. Retrieved 1 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Curetis Group Company Ares Genetics and BGI Group Collaborate to Offer Next-Generation Sequencing and PCR-based Coronavirus (2019-nCoV) Testing in Europe". GlobeNewswire News Room. 30 ខែមករា 2020. Archived from the original on 31 ខែមករា 2020. Retrieved 1 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Brueck, Hilary (30 ខែមករា 2020). "There's only one way to know if you have the coronavirus, and it involves machines full of spit and mucus". Business Insider. Archived from the original on 1 ខែកុម្ភៈ 2020. Retrieved 1 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases". Archived from the original on 21 ខែកុម្ភៈ 2020. Retrieved 26 ខែកុម្ភៈ 2020.
- ↑ (January 2020)"New SARS-like virus in China triggers alarm". Science 367 (6475): 234–35. DOI:10.1126/science.367.6475.234.
- ↑ "Severe acute respiratory syndrome coronavirus 2 data hub". NCBI. Archived from the original on 21 ខែមីនា 2020. Retrieved 4 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Petherick, Anna (4 April 2020). "Developing antibody tests for SARS-CoV-2". The Lancet 395 (10230): 1101–1102. ISSN 0140-6736. DOI:10.1016/s0140-6736(20)30788-1.
- ↑ Vogel, Gretchen (March 2020). "New blood tests for antibodies could show true scale of coronavirus pandemic". Science. DOI:10.1126/science.abb8028.
- ↑ (February 2020)"Potential Rapid Diagnostics, Vaccine and Therapeutics for 2019 Novel Coronavirus (2019-nCoV): A Systematic Review". Journal of Clinical Medicine 9 (3). DOI:10.3390/jcm9030623.
- ↑ AFP News Agency (11 April 2020). "How false negatives are complicating COVID-19 testing". Al Jazeera website Retrieved 12 April 2020.
- ↑ FDA (21 March 2020)។ "Coronavirus (COVID-19) Update: FDA Issues first Emergency Use Authorization for Point of Care Diagnostic"។ Press release។ https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-issues-first-emergency-use-authorization-point-care-diagnostic។ បានយកមក 22 March 2020។
- ↑ (February 2020)"A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (standard version)". Military Medical Research 7 (1). DOI:10.1186/s40779-020-0233-6.
- ↑ (24 February 2020)"COVID-19 pneumonia: what has CT taught us?". The Lancet Infectious Diseases 0 (4): 384–385. ISSN 1473-3099. DOI:10.1016/S1473-3099(20)30134-1.
- ↑ "ICD-10 Version:2019". World Health Organization. 2019. Archived from the original on 31 ខែមីនា 2020. Retrieved 31 ខែមីនា 2020.
U07.2—COVID-19, virus not identified—COVID-19 NOS—Use this code when COVID-19 is diagnosed clinically or epidemiologically but laboratory testing is inconclusive or not available. Use additional code, if desired, to identify pneumonia or other manifestations
- ↑ (March 2020)"Autopsy in suspected COVID-19 cases". J. Clin. Pathol. 73 (5): jclinpath-2020-206522. DOI:10.1136/jclinpath-2020-206522.
- ↑ (March 2020)"[A pathological report of three COVID-19 cases by minimally invasive autopsies]". Zhonghua Bing Li Xue Za Zhi 49: E009. DOI:10.3760/cma.j.cn112151-20200312-00193.
- ↑ (16 March 2020)"Exuberant plasmocytosis in bronchoalveolar lavage of the first patient requiring Extracorporeal Membrane Oxygenation for SARS-CoV-2 in Europe". Journal of Thoracic Oncology 15 (5): e65–e66. DOI:10.1016/j.jtho.2020.03.008.
- ↑ Lillicrap, David (1 April 2020). "Disseminated intravascular coagulation in patients with 2019-nCoV pneumonia". Journal of Thrombosis and Haemostasis: JTH 18 (4): 786–787. DOI:10.1111/jth.14781.
- ↑ (25 March 2020)"Leukoerythroblastic reaction in a patient with COVID-19 infection". American Journal of Hematology. DOI:10.1002/ajh.25793.
- ↑ Wiles, Siouxsie (9 ខែមីនា 2020). "The three phases of Covid-19—and how we can make it manageable". The Spinoff. Archived from the original on 27 ខែមីនា 2020. Retrieved 9 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ ១១៥,០ ១១៥,១ ១១៥,២ ១១៥,៣ (March 2020)"How will country-based mitigation measures influence the course of the COVID-19 epidemic?". Lancet 395 (10228): 931–934. DOI:10.1016/S0140-6736(20)30567-5.
- ↑ Barclay, Eliza (10 ខែមីនា 2020). "How canceled events and self-quarantines save lives, in one chart". Vox. Archived from the original on 12 ខែមីនា 2020. Retrieved 12 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Barclay, Eliza; Scott, Dylan; Animashaun, Animashaun (April 7, 2020). "The US doesn't just need to flatten the curve. It needs to "raise the line."". Vox. https://web.archive.org/web/20200407155950/https://www.vox.com/2020/4/7/21201260/coronavirus-usa-chart-mask-shortage-ventilators-flatten-the-curve.
- ↑ ១១៨,០ ១១៨,១ Wiles, Siouxsie (14 ខែមីនា 2020). "After 'Flatten the Curve', we must now 'Stop the Spread'. Here's what that means". The Spinoff. Archived from the original on 26 ខែមីនា 2020. Retrieved 13 ខែមីនា 2020.
- ↑ ១១៩,០ ១១៩,១ ១១៩,២ ១១៩,៣ Centers for Disease Control (3 ខែកុម្ភៈ 2020). "Coronavirus Disease 2019 (COVID-19): Prevention & Treatment". Archived from the original on 15 ខែធ្នូ 2019. Retrieved 10 ខែកុម្ភៈ 2020.
- ↑ World Health Organization. "Advice for Public". Archived from the original on 26 ខែមករា 2020. Retrieved 10 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "My Hand-Washing Song: Readers Offer Lyrics For A 20-Second Scrub". NPR.org. Archived from the original on 20 ខែមីនា 2020. Retrieved 20 ខែមីនា 2020.
- ↑ ១២២,០ ១២២,១ CDC (11 ខែកុម្ភៈ 2020). "Recommendation Regarding the Use of Cloth Face Coverings, Especially in Areas of Significant Community-Based Transmission". Centers for Disease Control and Prevention. Retrieved 17 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ ១២៣,០ ១២៣,១ ១២៣,២ ១២៣,៣ "COVID-19 Treatment Guidelines". www.nih.gov. National Institutes of Health. Archived from the original on 11 ខែតុលា 2020. Retrieved 21 ខែមេសា 2020.
- ↑ Maragakis, Lisa Lockerd. "Coronavirus, Social Distancing and Self Quarantine". www.hopkinsmedicine.org. Johns Hopkins University. Archived from the original on 18 ខែមីនា 2020. Retrieved 18 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Parker-Pope, Tara (19 March 2020). "Deciding How Much Distance You Should Keep". The New York Times. ល.ត.ម.អ. 0362-4331. https://www.nytimes.com/2020/03/19/well/live/coronavirus-quarantine-social-distancing.html.
- ↑ ១២៦,០ ១២៦,១ ១២៦,២ ១២៦,៣ ១២៦,៤ (April 13, 2020)"Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19): A Review". JAMA. DOI:10.1001/jama.2020.6019.
- ↑ Systrom, Kevin; Krieger, Mike; O'Rourke, Ryan; Stein, Robby; Dellaert, Frank; Lerer, Adam (11 ខែមេសា 2020). "Rt Covid-19". rt.live. Retrieved 19 ខែមេសា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) Based on (14 May 2008)"Real Time Bayesian Estimation of the Epidemic Potential of Emerging Infectious Diseases". PLOS ONE 3 (5): e2185. ISSN 1932-6203. DOI:10.1371/journal.pone.0002185. - ↑ Grenfell, Rob; Drew, Trevor (17 ខែកុម្ភៈ 2020). "Here's Why It's Taking So Long to Develop a Vaccine for the New Coronavirus". Science Alert. Archived from the original on 28 ខែកុម្ភៈ 2020. Retrieved 26 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "When and how to use masks". World Health Organization (WHO). Archived from the original on 7 ខែមីនា 2020. Retrieved 8 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Staff (8 ខែមេសា 2020). "Using face masks in the community—Technical Report" (PDF). ECDC.
{{cite web}}: CS1 maint: url-status (link) - ↑ "For different groups of people: how to choose masks". NHC.gov.cn. National Health Commission of the People's Republic of China. 7 ខែកុម្ភៈ 2020. Retrieved 22 ខែមីនា 2020.
Disposable medical masks: Recommended for: · People in crowded places · Indoor working environment with a relatively dense population · People going to medical institutions · Children in kindergarten and students at school gathering to study and do other activities
[តំណភ្ជាប់ខូច] - ↑ "Prevention of Coronavirus Disease 2019 (COVID-19)" (PDF). Centre for Health Protection. Archived from the original (PDF) on 21 ខែមីនា 2020. Retrieved 22 ខែមីនា 2020.
Wear a surgical mask when taking public transport or staying in crowded places.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help) - ↑ Staff (11 ខែមេសា 2020). "Spain officially recommends face mask use". Gazette Life. Retrieved 20 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ Giuffrida, Angela; Beaumont, Peter (2020-04-05). "Lombardy insists on face masks outside homes to stop Covid-19". The Guardian. ល.ត.ម.អ. 0261-3077. https://www.theguardian.com/world/2020/apr/05/lombardy-insists-on-face-masks-outside-homes-to-stop-covid-19.
- ↑ "Russian sanitary watchdog chief supports regions binding people to wear face masks". Tass. 2020-05-01. https://tass.com/society/1152425.
- ↑ "Coronavirus Disease 2019 (COVID-19)—Prevention & Treatment". Centers for Disease Control and Prevention. U.S. Department of Health & Human Services. 10 ខែមីនា 2020. Archived from the original on 11 ខែមីនា 2020. Retrieved 11 ខែមីនា 2020.
- ↑ "WHO-recommended handrub formulations". WHO Guidelines on Hand Hygiene in Health Care: First Global Patient Safety Challenge Clean Care Is Safer Care.. World Health Organization. 19 March 2009. https://www.ncbi.nlm.nih.gov/books/NBK144054/។ បានយកមក 19 March 2020.
- ↑ ១៣៨,០ ១៣៨,១ (February 2020)"Q&A: The novel coronavirus outbreak causing COVID-19". BMC Medicine 18 (1). DOI:10.1186/s12916-020-01533-w.
- ↑ (February 2020)"Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province". Chinese Medical Journal 133 (9): 1025–1031. DOI:10.1097/CM9.0000000000000744.
- ↑ (March 2020)"Comorbidities and multi-organ injuries in the treatment of COVID-19". Lancet 395 (10228): e52. DOI:10.1016/s0140-6736(20)30558-4.
- ↑ Henry, Brandon Michael (2020). "COVID-19, ECMO, and lymphopenia: a word of caution". The Lancet Respiratory Medicine 8 (4): e24. ISSN 2213-2600. DOI:10.1016/s2213-2600(20)30119-3.
- ↑ (19 March 2020)"A review of the 2019 novel coronavirus (COVID-19) based on current evidence". International Journal of Antimicrobial Agents. ISSN 0924-8579. DOI:10.1016/j.ijantimicag.2020.105948.
- ↑ (2020). "Unique epidemiological and clinical features of the emerging 2019 novel coronavirus pneumonia (COVID-19) implicate special control measures". Journal of Medical Virology n/a (n/a): 568–576. ISSN 1096-9071. DOI:10.1002/jmv.25748.
- ↑ (February 2020)"2019 Novel coronavirus: where we are and what we know". Infection 48 (2): 155–163. DOI:10.1007/s15010-020-01401-y.
- ↑ "Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected". World Health Organization (WHO). Archived from the original on 31 ខែមករា 2020. Retrieved 13 ខែកុម្ភៈ 2020.
- ↑ Farkas, Josh (March 2020) (digital). COVID-19—The Internet Book of Critical Care. USA: EMCrit. https://emcrit.org/ibcc/covid19/។ បានយកមក 13 March 2020.
- ↑ "COVID19—Resources for Health Care Professionals". Penn Libraries. 11 ខែមីនា 2020. Archived from the original on 14 ខែមីនា 2020. Retrieved 13 ខែមីនា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help) - ↑ "Frequently Asked Questions on the Emergency Use Authorization for Remdesivir for Certain Hospitalized COVID‐19 Patients" (PDF). U.S. Food and Drug Administration (FDA). 1 ខែឧសភា 2020. Retrieved 1 ខែឧសភា 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ↑ Day, Michael (17 March 2020). "Covid-19: ibuprofen should not be used for managing symptoms, say doctors and scientists". BMJ 368: m1086. ISSN 1756-1833. DOI:10.1136/bmj.m1086.
- ↑ "Self-isolation advice—Coronavirus (COVID-19)". National Health Service (United Kingdom). 28 ខែកុម្ភៈ 2020. Archived from the original on 28 ខែមីនា 2020. Retrieved 27 ខែមីនា 2020.
- ↑ Godoy, Maria (18 ខែមីនា 2020). "Concerned About Taking Ibuprofen For Coronavirus Symptoms? Here's What Experts Say". NPR. Retrieved 8 ខែមេសា 2020.
- ↑ AFP (19 ខែមីនា 2020). "Updated: WHO Now Doesn't Recommend Avoiding Ibuprofen For COVID-19 Symptoms". ScienceAlert. Archived from the original on 18 ខែមីនា 2020. Retrieved 19 ខែមីនា 2020.
- ↑ Research, Center for Drug Evaluation and (2020-03-19). "FDA advises patients on use of non-steroidal anti-inflammatory drugs (NSAIDs) for COVID-19". Drug Safety and Availability.
- ↑ "Patients taking ACE-i and ARBs who contract COVID-19 should continue treatment, unless otherwise advised by their physician". Archived from the original on 21 ខែមីនា 2020. Retrieved 21 ខែមីនា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help) - ↑ "Patients taking ACE-i and ARBs who contract COVID-19 should continue treatment, unless otherwise advised by their physician"។ Press release។ 17 March 2020។ https://newsroom.heart.org/news/patients-taking-ace-i-and-arbs-who-contract-covid-19-should-continue-treatment-unless-otherwise-advised-by-their-physician។ បានយកមក 25 March 2020។
- ↑ de Simone, Giovanni. "Position Statement of the ESC Council on Hypertension on ACE-Inhibitors and Angiotensin Receptor Blockers". Council on Hypertension of the European Society of Cardiology. Archived from the original on 24 ខែមីនា 2020. Retrieved 24 ខែមីនា 2020.
- ↑ "New Evidence Concerning Safety of ACE Inhibitors, ARBs in COVID-19". Pharmacy Times. Archived from the original on 11 ខែតុលា 2020. Retrieved 2 ខែឧសភា 2020.
- ↑ (February 2020)"Covid-19: a puzzle with many missing pieces". BMJ 368: m627. DOI:10.1136/bmj.m627.
- ↑ "Novel Coronavirus—COVID-19: What Emergency Clinicians Need to Know". www.ebmedicine.net. Archived from the original on 14 ខែមីនា 2020. Retrieved 9 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Grainger, Suzanne. "ASCIA Position Statement: Specific Treatments for COVID-19". Australasian Society of Clinical Immunology and Allergy (ASCIA) (in British English). Archived from the original on 16 ខែមិថុនា 2020. Retrieved 2 ខែឧសភា 2020.
- ↑ Tang, Ning (2020). "Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy". Journal of Thrombosis and Haemostasis n/a (n/a): 1094–1099. ISSN 1538-7836. DOI:10.1111/jth.14817.
- ↑ "Sequence for Putting On Personal Protective Equipment (PPE)" (PDF). CDC. Archived (PDF) from the original on 5 ខែមីនា 2020. Retrieved 8 ខែមីនា 2020.
- ↑ (February 2020)"Staff safety during emergency airway management for COVID-19 in Hong Kong". Lancet Respiratory Medicine 8 (4): e19. DOI:10.1016/s2213-2600(20)30084-9.
- ↑ "What healthcare personnel should know about caring for patients with confirmed or possible coronavirus disease 2" (PDF). CDC. 12 ខែមីនា 2020. Retrieved 31 ខែមីនា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ "Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention. 11 ខែកុម្ភៈ 2020. Archived from the original on 4 ខែមីនា 2020. Retrieved 11 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention. 11 ខែកុម្ភៈ 2020. Archived from the original on 4 ខែមីនា 2020. Retrieved 8 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ CDC (11 ខែកុម្ភៈ 2020). "Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings". Centers for Disease Control and Prevention. Archived from the original on 4 ខែមីនា 2020. Retrieved 25 ខែមីនា 2020.
- ↑ "Coronavirus Disease 2019 (COVID-19) Frequently Asked Questions". Food and Drug Administration.
- ↑ "Strategies for Optimizing the Supply of Facemasks". CDC. 11 ខែកុម្ភៈ 2020. Archived from the original on 23 ខែមីនា 2020. Retrieved 23 ខែមីនា 2020.
- ↑ ១៧០,០ ១៧០,១ ១៧០,២ (11 March 2020)"Care for Critically Ill Patients With COVID-19". JAMA 323 (15). DOI:10.1001/jama.2020.3633.
- ↑ World Health Organization (28 ខែមករា 2020). "Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected" (PDF). Archived (PDF) from the original on 26 ខែកុម្ភៈ 2020. Retrieved 18 ខែមីនា 2020.
- ↑ (30 March 2020)"The experience of high-flow nasal cannula in hospitalized patients with 2019 novel coronavirus-infected pneumonia in two hospitals of Chongqing, China". Annals of Intensive Care 10 (1). DOI:10.1186/s13613-020-00653-z.
- ↑ (April 2020)"COVID-19: Respiratory support outside the intensive care unit". The Lancet Respiratory Medicine. DOI:10.1016/S2213-2600(20)30176-4.
- ↑ (16 March 2020)"Report 9: Impact of non-pharmaceutical interventions (NPIs) to reduce COVID19 mortality and healthcare demand". DOI:10.25561/77482.
- ↑ ១៧៥,០ ១៧៥,១ Scott, Dylan (16 March 2020). "Coronavirus is exposing all of the weaknesses in the US health system High health care costs and low medical capacity made the US uniquely vulnerable to the coronavirus.". Vox. https://www.vox.com/policy-and-politics/2020/3/16/21173766/coronavirus-covid-19-us-cases-health-care-system.
- ↑ ១៧៦,០ ១៧៦,១ (March 2020)"Treatment for severe acute respiratory distress syndrome from COVID-19". The Lancet Respiratory Medicine 8 (5): 433–434. DOI:10.1016/S2213-2600(20)30127-2.
- ↑ (3 March 2010)"Higher vs Lower Positive End-Expiratory Pressure in Patients With Acute Lung Injury and Acute Respiratory Distress Syndrome". JAMA 303 (9): 865–73. DOI:10.1001/jama.2010.218.
- ↑ Diaz, Raiko; Heller, Daniel (2020). Barotrauma And Mechanical Ventilation. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK545226/.
- ↑ "Chinese doctors using plasma therapy on coronavirus, WHO says 'very valid' approach". Reuters. 17 February 2020. https://www.reuters.com/article/us-china-health-hospital-idUSKBN20B1M6.
- ↑ Steenhuysen, Julie; Kelland, Kate (24 January 2020). "With Wuhan virus genetic code in hand, scientists begin work on a vaccine". Reuters. https://www.reuters.com/article/us-china-health-vaccines-idUSKBN1ZN2J8.
- ↑ Duddu, Praveen (19 ខែកុម្ភៈ 2020). "Coronavirus outbreak: Vaccines/drugs in the pipeline for Covid-19". clinicaltrialsarena.com. Archived from the original on 19 ខែកុម្ភៈ 2020. Retrieved 7 ខែឧសភា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help); Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (28 January 2020)"Drug treatment options for the 2019-new coronavirus (2019-nCoV).". Biosci Trends 14 (1): 69–71. DOI:10.5582/bst.2020.01020.
- ↑ ១៨៣,០ ១៨៣,១ ១៨៣,២ ១៨៣,៣ (March 2020)"Therapeutic options for the 2019 novel coronavirus (2019-nCoV)". Nature Reviews. Drug Discovery 19 (3): 149–150. DOI:10.1038/d41573-020-00016-0.
- ↑ Nebehay, Stephanie; Kelland, Kate; Liu, Roxanne (5 February 2020). "WHO: 'no known effective' treatments for new coronavirus". Thomson Reuters. https://www.reuters.com/article/us-china-health-treatments-who-idUSKBN1ZZ1M6.
- ↑ "Blood plasma taken from covid-19 survivors might help patients fight off the disease". MIT Technology Review.
- ↑ "Trials of Plasma From Recovered Covid-19 Patients Have Begun". Wired.
- ↑ "FDA now allows treatment of life-threatening COVID-19 cases using blood from patients who have recovered". TechCrunch. Archived from the original on 11 ខែតុលា 2020. Retrieved 9 ខែមេសា 2020.
- ↑ "China launches coronavirus 'close contact' app". BBC News. 11 February 2020. https://www.bbc.com/news/technology-51439401.
- ↑ Chen, Angela. "China's coronavirus app could have unintended consequences". MIT Technology Review. Retrieved 7 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ "Gov in the Time of Corona". GovInsider. 19 March 2020. https://govinsider.asia/innovation/gov-in-the-time-of-corona/.
- ↑ Manancourt, Vincent (10 March 2020). "Coronavirus tests Europe's resolve on privacy". POLITICO. https://www.politico.eu/article/coronavirus-tests-europe-resolve-on-privacy-tracking-apps-germany-italy/.
- ↑ Tidy, Joe (17 March 2020). "Coronavirus: Israel enables emergency spy powers". BBC News. https://www.bbc.com/news/technology-51930681.
- ↑ Bünte, Oliver (18 March 2020). "Corona-Krise: Deutsche Telekom liefert anonymisierte Handydaten an RKI" (ជាGerman). Heise Online. https://www.heise.de/newsticker/meldung/Corona-Krise-Deutsche-Telekom-liefert-anonymisierte-Handydaten-an-RKI-4685191.html.
- ↑ "Moscow deploys facial recognition technology for coronavirus quarantine". Reuters. 21 February 2020. https://www.reuters.com/article/us-china-health-moscow-technology-idUSKBN20F1RZ.
- ↑ "Italians scolded for flouting lockdown as death toll nears 3,000". Pittsburgh Post-Gazette. https://www.post-gazette.com/news/world/2020/03/18/Italy-coronavirus-475-deaths-one-day-death-toll-2978-COVID-19-doctors/stories/202003180182.
- ↑ "Kreative Lösungen gesucht". Startseite (in អាល្លឺម៉ង់). Archived from the original on 24 ខែមីនា 2020. Retrieved 23 ខែមីនា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help) - ↑ Dannewitz, Juliane (23 March 2020). "Hackathon Germany: #WirvsVirus" (ជាde-DE). Datenschutzbeauftragter. https://www.datenschutzbeauftragter-info.de/hackathon-germany-wirvsvirus/.
- ↑ Staff (8 ខែមេសា 2020). "Lockdown is working, suggests latest data from symptom tracker app". Kings College London News Centre. Retrieved 20 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ Whyte, Andrew (21 March 2020). "President makes global call to combat coronavirus via hackathon". ERR. https://news.err.ee/1067171/president-makes-global-call-to-combat-coronavirus-via-hackathon.
- ↑ (March 2020)"Timely mental health care for the 2019 novel coronavirus outbreak is urgently needed". The Lancet. Psychiatry 7 (3): 228–29. DOI:10.1016/S2215-0366(20)30046-8.
- ↑ (March 2020)"The mental health of medical workers in Wuhan, China dealing with the 2019 novel coronavirus". The Lancet. Psychiatry 7 (3): e14. DOI:10.1016/S2215-0366(20)30047-X.
- ↑ Coronavirus: 'Profound' mental health impact prompts calls for urgent research, BBC, Philippa Roxby, April 16, 2020.
- ↑ Multidisciplinary research priorities for the COVID‑19 pandemic: a call for action for mental health science, The Lancet, Emily Holmes, Rory O'Connor, Hugh Perry, et al., April 15, 2020, page 1: "A fragmented research response, characterised by small-scale and localised initiatives, will not yield the clear insights necessary to guide policymakers or the public."
- ↑ (4 March 2020)"Coronavirus Disease (COVID-19)". Our World in Data.
- ↑ ២០៥,០ ២០៥,១ ២០៥,២ ២០៥,៣ (17 February 2020)"The Epidemiological Characteristics of an Outbreak of 2019 Novel Coronavirus Diseases (COVID-19)—China, 2020". China CDC Weekly 2 (8): 113–122.
- ↑ (ជាko) 코로나바이러스감염증-19 국내 발생 현황(7월 17일, 정례브리핑) (Report). Korea Centers for Disease Control and Prevention. 2020-07-17. https://www.cdc.go.kr/board/board.es?mid=a20501000000&bid=0015&list_no=367829&act=view។ បានយកមក 2020-07-17.
- ↑ ២០៧,០ ២០៧,១ (ជាSpanish) Actualización nº 109. Enfermedad por el coronavirus (COVID-19) (Report). Ministerio de Sanidad, Consumo y Bienestar Social. 18 May 2020. https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov-China/documentos/Actualizacion_109_COVID-19.pdf។ បានយកមក 20 May 2020. Cite error: Invalid
<ref>tag; name "MSCBSReport" defined multiple times with different content - ↑ "Epidemia COVID-19 – Bollettino sorveglianza integrata COVID-19" (PDF) (in អ៊ីតាលី). Istituto Superiore di Sanità. 5 ខែមិថុនា 2020. Retrieved 10 ខែមិថុនា 2020.
- ↑ (6 April 2020)"Coronavirus Disease (COVID-19)". Our World in Data.
- ↑ ២១០,០ ២១០,១ (April 2020)"Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection in Children and Adolescents A Systematic Review". JAMA Pediatrics. DOI:10.1001/jamapediatrics.2020.1467. Retrieved on 22 April 2020.
- ↑ (18 March 2020)"SARS-CoV-2 Infection in Children". New England Journal of Medicine 382 (17): 1663–1665. ISSN 0028-4793. DOI:10.1056/nejmc2005073.
- ↑ (2020)"Epidemiological Characteristics of 2143 Pediatric Patients With 2019 Coronavirus Disease in China". Pediatrics: e20200702. DOI:10.1542/peds.2020-0702.
- ↑ (March 2020)"Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection?". The Lancet Respiratory Medicine 395 (10224): e40. DOI:10.1016/S0140-6736(20)30311-1.
- ↑ "Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention. 11 ខែកុម្ភៈ 2020. Archived from the original on 2 ខែមីនា 2020. Retrieved 2 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (12 March 2020)"Dysregulation of immune response in patients with COVID-19 in Wuhan, China". Clinical Infectious Diseases. DOI:10.1093/cid/ciaa248.
- ↑ "WHO Director-General's statement on the advice of the IHR Emergency Committee on Novel Coronavirus". World Health Organization (WHO).
- ↑ ២១៧,០ ២១៧,១ Characteristics of COVID-19 patients dying in Italy Report based on available data on April 2th, 2020 (Report). Istituto Superiore di Sanità. 3 April 2020. https://www.epicentro.iss.it/en/coronavirus/bollettino/Report-COVID-2019_2_april_2020.pdf។ បានយកមក 3 April 2020.
- ↑ (April 2020)"Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan, China". Journal of Medical Virology 92 (4): 441–47. DOI:10.1002/jmv.25689.
- ↑ "Coronavirus Age, Sex, Demographics (COVID-19)". www.worldometers.info. Archived from the original on 27 ខែកុម្ភៈ 2020. Retrieved 26 ខែកុម្ភៈ 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (2020-04-08)"Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019—COVID-NET, 14 States, March 1–30, 2020". MMWR. Morbidity and Mortality Weekly Report 69 (15): 458–464. ISSN 0149-2195. DOI:10.15585/mmwr.mm6915e3.
- ↑ (February 2020)"Potential association between COVID-19 mortality and health-care resource availability". Lancet Global Health 8 (4): e480. DOI:10.1016/S2214-109X(20)30068-1.
- ↑ (March 2020)"[Comparison of epidemic characteristics between SARS in2003 and COVID-19 in 2020 in Guangzhou]". Zhonghua Liu Xing Bing Xue Za Zhi = Zhonghua Liuxingbingxue Zazhi 41 (5): 634–637. DOI:10.3760/cma.j.cn112338-20200228-00209.
- ↑ (February 2020)"Real-Time Estimation of the Risk of Death from Novel Coronavirus (COVID-19) Infection: Inference Using Exported Cases". Journal of Clinical Medicine 9 (2). DOI:10.3390/jcm9020523.
- ↑ (March 2020)"Is Coronavirus disease (COVID-19) case fatality ratio underestimated?". Global Biosecurity 1 (3). DOI:10.31646/gbio.56.
- ↑ (March 2020)"Real estimates of mortality following COVID-19 infection.". The Lancet Infectious Diseases. DOI:10.1016/S1473-3099(20)30195-X.
- ↑ (20 March 2020)"COVID-19 and smoking: A systematic review of the evidence". Tobacco Induced Diseases 18 (March). ISSN 1617-9625. DOI:10.18332/tid/119324.
- ↑ Cheung, Elizabeth (13 ខែមីនា 2020). "Some recovered Covid-19 patients may have lung damage, doctors say". South China Morning Post. Archived from the original on 15 ខែមីនា 2020. Retrieved 15 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ Servick, Kelly (2020-04-08). "For survivors of severe COVID-19, beating the virus is just the beginning". Science. ISSN 0036-8075. DOI:10.1126/science.abc1486.
- ↑ "Coronavirus Disease 2019 (COVID-19) DAILY EPIDEMIOLOGY UPDATE Updated: 5 May, 2020, 11:00 AM ET" (PDF). Public Health Agency of Canada. 5 ខែឧសភា 2020. Retrieved 6 ខែឧសភា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ (ជាDanish) COVID-19 i Danmark: Epidemiologisk overvågningsrapport den 5. maj 2020 (Report). Statens Serum Institut. 2020-05-05. បានដាក់ទុកឯកសារ ពី[១] នៅថ្ងៃ 10 ឧសភា 2020. https://web.archive.org/web/20200510173051/https://files.ssi.dk/COVID19-overvaagningsrapport-30042020-2h7d។ បានយកមក 2020-05-05.
- ↑ https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Situationsberichte/2020-05-05-de.pdf
- ↑ "Robert Koch-Institut: COVID-19-Dashboard". Experience.arcgis.com. Retrieved 7 ខែឧសភា 2020.
- ↑ "קורונה - משרד הבריאות". Ministry of Health (Israel). 3 ខែឧសភា 2020. Retrieved 5 ខែឧសភា 2020.
- ↑ (ជាItalian) Epidemia COVID-19. Aggiornamento nazionale 28 aprile 2020 (Report). Rome: Istituto Superiore di Sanità. 29 April 2020. https://www.epicentro.iss.it/coronavirus/bollettino/Bollettino-sorveglianza-integrata-COVID-19_28-aprile-2020.pdf។ បានយកមក 30 April 2020.
- ↑ "KDPH COVID-19 Dashboard". Kygeonet.maps.arcgis.com. Retrieved 7 ខែឧសភា 2020.
- ↑ "COVID-19 Response Reporting". Mass.gov. 1 ខែមករា 2020. Retrieved 7 ខែឧសភា 2020.
- ↑ (ជាDutch) Epidemiologische situatie COVID-19 in Nederland 25 april 2020 (Report). Bilthoven: Rijksinstituut voor Volksgezondheid en Milie. 25 April 2020. https://www.rivm.nl/documenten/epidemiologische-situatie-covid-19-in-nederland-25-april-2020។ បានយកមក 25 April 2020.
- ↑ https://www.fhi.no/contentassets/ca5914bd0aa14e15a17f8a7d48fa306a/2020_05_05_dagsrapport-norge-covid-19.pdf[តំណភ្ជាប់ខូច]
- ↑ "NOVO CORONAVÍRUS COVID-19 RELATÓRIO DE SITUAÇÃO" (PDF) (in ព័រទុយហ្គាល់). 5 ខែឧសភា 2020. Archived from the original (PDF) on 5 ខែឧសភា 2020. Retrieved 5 ខែឧសភា 2020.
- ↑ (ជាko) 코로나바이러스감염증-19 국내 발생 현황(4월 30일, 정례브리핑) (Report). Korea Centers for Disease Control and Prevention. 30 April 2020. https://www.cdc.go.kr/board/board.es?mid=a20501000000&bid=0015&list_no=367051&act=view។ បានយកមក 30 April 2020.
- ↑ "FOHM Covid-19". Public Health Agency of Sweden. Retrieved 5 ខែឧសភា 2020.
- ↑ "Todesfälle in der Schweiz nach Altersgruppen". datawrapper.dwcdn.net. 30 ខែមេសា 2020. Retrieved 30 ខែមេសា 2020.
- ↑ "2019 Novel Coronavirus Outbreak (COVID-19)". Washington State Department of Health. 5 ខែឧសភា 2020. Retrieved 6 ខែឧសភា 2020.
- ↑ CDC COVID-19 Response Team (18 March 2020). "Severe Outcomes Among Patients with Coronavirus Disease 2019 (COVID-19)—United States, February 12 – March 16, 2020". Morbidity and Mortality Weekly Report 69 (12): 343–346. DOI:10.15585/mmwr.mm6912e2.
- ↑ Verity, Robert (30 March 2020). "Estimates of the severity of coronavirus disease 2019: a model-based analysis". The Lancet Infectious Diseases. ISSN 1473-3099. DOI:10.1016/s1473-3099(20)30243-7.
- ↑ "BSI open letter to Government on SARS-CoV-2 outbreak response". immunology.org. British Society for Immunology. Archived from the original on 14 ខែមីនា 2020. Retrieved 15 ខែមីនា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help) - ↑ Schraer, Rachel (25 April 2020). "Coronavirus: Immunity passports 'could increase virus spread'". https://www.bbc.com/news/world-52425825។ បានយកមក 26 April 2020.
- ↑ "Can you get coronavirus twice or does it cause immunity?". 13 March 2020. https://www.independent.co.uk/life-style/health-and-families/coronavirus-immunity-reinfection-get-covid-19-twice-sick-spread-relapse-a9400691.html.
- ↑ Politi, Daniel (11 April 2020). "WHO Investigating Reports of Coronavirus Patients Testing Positive Again After Recovery". Slate. https://slate.com/news-and-politics/2020/04/who-reports-coronavirus-testing-positive-recovery.html។ បានយកមក 11 April 2020.
- ↑ "They survived the coronavirus. Then they tested positive again. Why?". Los Angeles Times. 13 ខែមីនា 2020. Archived from the original on 14 ខែមីនា 2020. Retrieved 15 ខែមីនា 2020.
- ↑ "14% of Recovered Covid-19 Patients in Guangdong Tested Positive Again". caixinglobal.com. Caixin Global. Archived from the original on 3 ខែមីនា 2020. Retrieved 15 ខែមីនា 2020.
- ↑ ២៥២,០ ២៥២,១ (6 April 2020)"The COVID-19 Pandemic in the US A Clinical Update". JAMA. DOI:10.1001/jama.2020.5788.
- ↑ Richard Lloyd Parry (៣០ ខែមេសា ២០២០), "Coronavirus patients can't relapse, South Korean scientists believe", The Times, https://www.thetimes.co.uk/article/coronavirus-patients-cant-relapse-south-korean-scientists-believe-rkm8zm7d9
- ↑ "What if immunity to covid-19 doesn't last?". MIT Technology Review (in អង់គ្លេស). Retrieved 1 ខែឧសភា 2020.
- ↑ "Direct observation of repeated infections with endemic coronaviruses" (PDF). Columbia University in the City of New York (in អង់គ្លេស). Department of Environmental Health Sciences, Mailman School of Public Health, Columbia University. 15 ខែមេសា 2020. Retrieved 2 ខែឧសភា 2020.
- ↑ Berger, Kevin (12 ខែមីនា 2020). "The Man Who Saw the Pandemic Coming". Nautilus. Archived from the original on 15 ខែមីនា 2020. Retrieved 16 ខែមីនា 2020.
{{cite web}}: More than one of|archivedate=and|archive-date=specified (help); More than one of|archiveurl=and|archive-url=specified (help); Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (February 2020)"COVID-19: what is next for public health?". Lancet 395 (10224): 542–45. DOI:10.1016/S0140-6736(20)30374-3.
- ↑ Wu, Yi-Chi (March 2020). "The outbreak of COVID-19: An overview". Journal of the Chinese Medical Association 83 (3): 217–220. ISSN 1726-4901. DOI:10.1097/JCMA.0000000000000270.
- ↑ (February 2020)"A novel coronavirus outbreak of global health concern". Lancet 395 (10223): 470–473. DOI:10.1016/S0140-6736(20)30185-9.
- ↑ Cohen, Jon (January 2020). "Wuhan seafood market may not be source of novel virus spreading globally". Science. DOI:10.1126/science.abb0611.
- ↑ "Novel Coronavirus—China". World Health Organization (WHO). 12 ខែមករា 2020.
- ↑ Kessler, Glenn (17 April 2020). "Trump's false claim that the WHO said the coronavirus was 'not communicable'". The Washington Post. https://archive.today/20200417193804/https://www.washingtonpost.com/politics/2020/04/17/trumps-false-claim-that-who-said-coronavirus-was-not-communicable/។ បានយកមក 17 April 2020.
- ↑ Kuo, Lily (21 January 2020). "China confirms human-to-human transmission of coronavirus". The Guardian. https://www.theguardian.com/world/2020/jan/20/coronavirus-spreads-to-beijing-as-china-confirms-new-cases។ បានយកមក 18 April 2020.
- ↑ "Principles of Epidemiology | Lesson 3—Section 3". www.cdc.gov. 18 ខែកុម្ភៈ 2019. Archived from the original on 28 ខែកុម្ភៈ 2020. Retrieved 28 ខែមីនា 2020.
- ↑ Ritchie, Hannah; Roser, Max (25 ខែមីនា 2020). Chivers, Tom (ed.). "What do we know about the risk of dying from COVID-19?". Our World in Data. Archived from the original on 28 ខែមីនា 2020. Retrieved 28 ខែមីនា 2020.
- ↑ Lazzerini, Marzia (18 March 2020). "COVID-19 in Italy: momentous decisions and many uncertainties". The Lancet Global Health 0 (5): e641–e642. ISSN 2214-109X. DOI:10.1016/S2214-109X(20)30110-8.
- ↑ "What do we know about the risk of dying from COVID-19?". Our World in Data. Archived from the original on 28 ខែមីនា 2020. Retrieved 28 ខែមីនា 2020.
- ↑ ២៦៨,០ ២៦៨,១ (April 2020)"COVID-19 in Prisons and Jails in the United States.". JAMA Internal Medicine. DOI:10.1001/jamainternmed.2020.1856.
- ↑ "Total confirmed cases of COVID-19 per million people". Our World in Data. Archived from the original on 19 ខែមីនា 2020. Retrieved 10 ខែមេសា 2020.
- ↑ "Total confirmed deaths due to COVID-19 per million people". Our World in Data. Archived from the original on 19 ខែមីនា 2020. Retrieved 10 ខែមេសា 2020.
- ↑ "What do we know about the risk of dying from COVID-19?". Our World in Data. Retrieved 23 ខែមេសា 2020.
- ↑ "Coronavirus disease 2019 (COVID-19) Situation Report—31" (PDF). 20 ខែកុម្ភៈ 2020. Retrieved 23 ខែមេសា 2020.
- ↑ ២៧៣,០ ២៧៣,១ "Global Covid-19 Case Fatality Rates". Centre for Evidence-Based Medicine. 17 ខែមីនា 2020. Archived from the original on 18 ខែឧសភា 2020. Retrieved 10 ខែមេសា 2020.
- ↑ Haake, Daniel (24 ខែមេសា 2020). "Gangelt — A representative study on the lethality of COVID-19". Medium. Retrieved 27 ខែមេសា 2020.[តំណភ្ជាប់ខូច]
- ↑ VogelApr. 21, Gretchen; 2020; Pm, 6:30 (21 ខែមេសា 2020). "Antibody surveys suggesting vast undercount of coronavirus infections may be unreliable". Science | AAAS. Retrieved 29 ខែមេសា 2020.
{{cite web}}: CS1 maint: numeric names: authors list (link) - ↑ Modi, Chirag (21 ខែមេសា 2020). "How deadly is COVID-19? Data Science offers answers from Italy mortality data". Medium. Retrieved 23 ខែមេសា 2020.
- ↑ Modi, Chirag (2020-04-20). "Total COVID-19 Mortality in Italy: Excess Mortality and Age Dependence through Time-Series Analysis". MedRxiv: 2020.04.15.20067074. DOI:10.1101/2020.04.15.20067074.
- ↑ Wu, Jin; McCann, Allison; Katz, Josh; Peltier, Elian (2020-04-27). "36,000 Missing Deaths: Tracking the True Toll of the Coronavirus Crisis". The New York Times. ល.ត.ម.អ. 0362-4331. https://www.nytimes.com/interactive/2020/04/21/world/coronavirus-missing-deaths.html.
- ↑ "The Coronavirus Isn't Just the Flu, Bro". www.bloomberg.com. Retrieved 26 ខែមេសា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ Mole, Beth (24 ខែមេសា 2020). "Experts demolish studies suggesting COVID-19 is no worse than flu". Ars Technica. Retrieved 26 ខែមេសា 2020.
- ↑ ២៨១,០ ២៨១,១ ២៨១,២ (14 March 2020)"COVID-19: the gendered impacts of the outbreak". The Lancet 395 (10227): 846–848. DOI:10.1016/S0140-6736(20)30526-2.
- ↑ (17 February 2020)"The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China". Chinese Journal of Epidemiology 41 (2): 145–151. ISSN 0254-6450. DOI:10.3760/cma.j.issn.0254-6450.2020.02.003. Retrieved on 7 April 2020.
- ↑ ២៨៣,០ ២៨៣,១ ២៨៣,២ ២៨៣,៣ Rabin, Roni Caryn (20 March 2020). "In Italy, Coronavirus Takes a Higher Toll on Men". The New York Times. https://www.nytimes.com/2020/03/20/health/coronavirus-italy-men-risk.html។ បានយកមក 7 April 2020.
- ↑ "COVID-19 weekly surveillance report". www.euro.who.int. Archived from the original on 15 ខែមីនា 2020. Retrieved 7 ខែមេសា 2020.
- ↑ ២៨៥,០ ២៨៥,១ Gupta, Alisha Haridasani (3 April 2020). "Does Covid-19 Hit Women and Men Differently? U.S. Isn't Keeping Track". The New York Times. https://www.nytimes.com/2020/04/03/us/coronavirus-male-female-data-bias.html។ បានយកមក 7 April 2020.
- ↑ "Gender equity in the health workforce: Analysis of 104 countries" (PDF). World Health Organization. Retrieved 7 ខែមេសា 2020.
- ↑ "Gender and the Coronavirus Outbreak: Think Global Health". Council on Foreign Relations. Retrieved 7 ខែមេសា 2020.
- ↑ ២៨៨,០ ២៨៨,១ (April 2020)"COVID-19 exacerbating inequalities in the US". The Lancet 395 (10232): 1243–1244. DOI:10.1016/S0140-6736(20)30893-X.
- ↑ (August 2020)"Population-Based Estimates of Chronic Conditions Affecting Risk for Complications from Coronavirus Disease, United States". Emerging Infectious Diseases 26 (8). DOI:10.3201/eid2608.200679.
- ↑ (18 April 2020)"The COVID-19 Pandemic: a Call to Action to Identify and Address Racial and Ethnic Disparities". Journal of Racial and Ethnic Health Disparities. DOI:10.1007/s40615-020-00756-0.
- ↑ CDC (11 ខែកុម្ភៈ 2020). "Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention (in American English). Retrieved 4 ខែឧសភា 2020.
- ↑ DeRobertis, Jacqueline (3 ខែឧសភា 2020). "People who use drugs are more vulnerable to coronavirus. Here's what clinics are doing to help". The Advocate (Louisiana) (in អង់គ្លេស). Retrieved 4 ខែឧសភា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ "2nd U.S. Case Of Wuhan Coronavirus Confirmed". NPR.org. Retrieved 4 ខែមេសា 2020.
- ↑ Jr, Donald G. McNeil (2020-02-02). "Wuhan Coronavirus Looks Increasingly Like a Pandemic, Experts Say". The New York Times. ល.ត.ម.អ. 0362-4331. https://www.nytimes.com/2020/02/02/health/coronavirus-pandemic-china.html.
- ↑ Griffiths, James. "Wuhan coronavirus deaths spike again as outbreak shows no signs of slowing". https://www.cnn.com/2020/02/05/asia/wuhan-coronavirus-update-death-toll-spike-intl-hnk/index.html.
- ↑ (February 2020)"A novel coronavirus (2019-nCoV) causing pneumonia-associated respiratory syndrome". Cellular & Molecular Immunology 17 (554). DOI:10.1038/s41423-020-0372-4.
- ↑ (February 2020)"A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster". Lancet 395 (10223): 514–523. DOI:10.1016/S0140-6736(20)30154-9.
- ↑ (22 September 2017)"The legacy of the Spanish flu". Science 357 (6357). ISSN 0036-8075. DOI:10.1126/science.aao4093.
- ↑ "Stop the coronavirus stigma now". Nature: p. 165. 7 April 2020. អ.វ.ល.:10.1038/d41586-020-01009-0. https://www.nature.com/articles/d41586-020-01009-0។ បានយកមក 16 April 2020.
- ↑ "Novel Coronavirus (2019-nCoV) SITUATION REPORT—1" (PDF). WHO. 21 ខែមករា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ "Novel Coronavirus(2019-nCoV) Situation Report—10" (PDF). WHO. 30 ខែមករា 2020.
{{cite web}}: CS1 maint: url-status (link) - ↑ "Novel coronavirus named 'Covid-19': WHO". TODAYonline. https://www.todayonline.com/world/wuhan-novel-coronavirus-named-covid-19-who.
- ↑ "The coronavirus spreads racism against—and among—ethnic Chinese". The Economist. 17 February 2020. https://www.economist.com/china/2020/02/17/the-coronavirus-spreads-racism-against-and-among-ethnic-chinese.
- ↑ "World Health Organization Best Practices for the Naming of New Human Infectious Diseases" (PDF). WHO. ខែឧសភា 2015.
{{cite web}}: CS1 maint: url-status (link) - ↑ ៣០៥,០ ៣០៥,១ "Naming the coronavirus disease (COVID-19) and the virus that causes it". World Health Organization (WHO). Archived from the original on 28 ខែកុម្ភៈ 2020. Retrieved 13 ខែមីនា 2020.
- ↑ "Coronavirus disease 2019 (COVID-19) in the EU/EEA and the UK—eighth update". ecdc. https://www.ecdc.europa.eu/sites/default/files/documents/covid-19-rapid-risk-assessment-coronavirus-disease-2019-eighth-update-8-april-2020.pdf.
- ↑ "China coronavirus: Misinformation spreads online about origin and scale". BBC News. 30 January 2020. https://www.bbc.com/news/blogs-trending-51271037.
- ↑ ទំព័រគំរូ:Cite newspaper
- ↑ "Here's A Running List Of Disinformation Spreading About The Coronavirus". Buzzfeed News. Archived from the original on 6 ខែកុម្ភៈ 2020. Retrieved 8 ខែកុម្ភៈ 2020.
- ↑ (16 April 2020)"Decline of acute coronary syndrome admissions in Austria since the outbreak of COVID-19: the pandemic response causes cardiac collateral damage". European Heart Journal. DOI:10.1093/eurheartj/ehaa314.
- ↑ (April 2020)"PRE-PRINT: Reduction in ST-Segment Elevation Cardiac Catheterization Laboratory Activations in the United States during COVID-19 Pandemic". Journal of the American College of Cardiology (JACC). DOI:10.1016/j.jacc.2020.04.011.
- ↑ ៣១២,០ ៣១២,១ 'Where are all our patients?': Covid phobia is keeping people with serious heart symptoms away from ERs, Stat News, Usha Lee McFarling, April 23, 2020.
- ↑ See also . . Amid the Coronavirus Crisis, Heart and Stroke Patients Go Missing, New York Times, Gina Kolata, April 25, 2020.
- ↑ "Coronavirus: Belgian cat infected by owner". Brusselstimes.com. 27 ខែមីនា 2020. Retrieved 12 ខែមេសា 2020.
- ↑ Goldstein, Joseph (6 April 2020). "Bronx Zoo Tiger Is Sick With the Coronavirus". https://www.nytimes.com/2020/04/06/nyregion/bronx-zoo-tiger-coronavirus.html.
- ↑ "Coronavirus hits Netherlands farm animals as minks test positive for virus". Fox News. 26 ខែមេសា 2020. Retrieved 27 ខែមេសា 2020.
- ↑ (8 April 2020)"Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS–coronavirus 2". Science: eabb7015. DOI:10.1126/science.abb7015.
- ↑ (March 2020)"COVID-19, an emerging coronavirus infection: advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics". Human Vaccines and Immunotherapeutics: 1–7. ISSN 2164-5515. DOI:10.1080/21645515.2020.1735227.
- ↑ (March 2020)"Potential interventions for novel coronavirus in China: A systematic review". Journal of Medical Virology 92 (5): 479–90. ISSN 0146-6615. DOI:10.1002/jmv.25707.
- ↑ ៣២០,០ ៣២០,១ (22 March 2020)"WHO launches global megatrial of the four most promising coronavirus treatments". Science Magazine. DOI:10.1126/science.abb8497.
- ↑ Bradley-Ridout, Glyneva; Fuller, Kaitlin; Gray, Mikaela; Nekolaichuk, Erica (9 ខែមេសា 2020). "Navigating the COVID-19 Evidence Landscape". University of Toronto Libraries - Gerstein Science Information Centre.
{{cite web}}: CS1 maint: url-status (link) - ↑ "Features, Evaluation and Treatment Coronavirus (COVID-19)". StatPearls [Internet]. StatPearls. March 2020. Bookshelf ID: NBK554776. https://www.ncbi.nlm.nih.gov/books/NBK554776/.
- ↑ (3 March 2020)"The SARS-CoV-2 Vaccine Pipeline: an Overview". Current Tropical Medicine Reports: 1–4. DOI:10.1007/s40475-020-00201-6.
- ↑ Roberts, Michelle (17 March 2020). "Coronavirus: US volunteers test first vaccine". BBC News. https://www.bbc.com/news/health-51906604.
- ↑ (2020). "News Feature: Avoiding pitfalls in the pursuit of a COVID-19 vaccine". Proceedings of the National Academy of Sciences of the United States of America 117 (15): 8218–8221. DOI:10.1073/pnas.2005456117.
- ↑ ៣២៦,០ ៣២៦,១ ៣២៦,២ "COVID-19 treatment and vaccine tracker" (PDF). Milken Institute. 21 ខែមេសា 2020. Retrieved 21 ខែមេសា 2020. Lay summary.
{{cite web}}: Cite uses deprecated parameter|lay-url=(help) - ↑ ៣២៧,០ ៣២៧,១ ៣២៧,២ ៣២៧,៣ Selina Koch; Winnie Pong (13 ខែមីនា 2020). "First up for COVID-19: nearly 30 clinical readouts before end of April". BioCentury Inc. Retrieved 1 ខែមេសា 2020.
- ↑ COVID-19 Clinical Research Coalition (2020). "Global coalition to accelerate COVID-19 clinical research in resource-limited settings". The Lancet 395 (10233): 1322–1325. ISSN 0140-6736. DOI:10.1016/s0140-6736(20)30798-4.
- ↑ Maguire, Brittany J. (2 April 2020). "A living systematic review protocol for COVID-19 clinical trial registrations". Wellcome Open Research 5. ISSN 2398-502X. DOI:10.12688/wellcomeopenres.15821.1.
- ↑ "UN health chief announces global 'solidarity trial' to jumpstart search for COVID-19 treatment". UN News. 18 ខែមីនា 2020. Archived from the original on 23 ខែមីនា 2020. Retrieved 23 ខែមីនា 2020.
- ↑ (March 2020)"Arguments in favor of remdesivir for treating SARS-CoV-2 infections". International Journal of Antimicrobial Agents 55 (4). DOI:10.1016/j.ijantimicag.2020.105933.
- ↑ Feuerstein, Adam; Garde, Damian; Robbins, Rebecca (16 ខែមេសា 2020). "Gilead data suggests coronavirus patients are responding to treatment". STAT. Retrieved 16 ខែមេសា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ↑ (April 2020)"Compassionate Use of Remdesivir for Patients with Severe Covid-19". N. Engl. J. Med.. DOI:10.1056/NEJMoa2007016.
- ↑ ៣៣៤,០ ៣៣៤,១ (February 2020)"Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro". Cell Research 30 (3): 269–71. DOI:10.1038/s41422-020-0282-0.
- ↑ (2020)"BMJ Best Practices: COVID-19". BMJ.
- ↑ (March 2020)"A Trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19". New England Journal of Medicine. DOI:10.1056/NEJMoa2001282.
- ↑ Seley-Radtke, Katherine (3 April 2020). "Professor of Chemistry and Biochemistry and President-Elect of the International Society for Antiviral Research, University of Maryland, Baltimore County". The Conversation. https://theconversation.com/a-small-trial-finds-that-hydroxychloroquine-is-not-effective-for-treating-coronavirus-135484.
- ↑ Molina, Jean Michel (March 2020). "No Evidence of Rapid Antiviral Clearance or Clinical Benefit with the Combination of Hydroxychloroquine and Azithromycin in Patients with Severe COVID-19 Infection". Médecine et Maladies Infectieuses: S0399077X20300858. DOI:10.1016/j.medmal.2020.03.006.
- ↑ (March 2020)"In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)". Clin. Infect. Dis.. DOI:10.1093/cid/ciaa237.
- ↑ Liu, Roxanne; Miller, Josh (3 March 2020). "China approves use of Roche drug in battle against coronavirus complications". Reuters. https://www.reuters.com/article/us-health-coronavirus-china-roche-hldg/china-approves-use-of-roche-arthritis-drug-for-coronavirus-patients-idUSKBN20R0LF.
- ↑ (5 March 2020)"Effective Treatment of Severe COVID-19 Patients with Tocilizumab". DOI:10.12074/202003.00026.
- ↑ Ovadia, Daniela; Agenzia, Zoe. "COVID-19—Italy launches an independent trial on tocilizumab". Univadis from Medscape. Aptus Health. Retrieved 22 ខែមេសា 2020.
- ↑ "Tocilizumab in COVID-19 Pneumonia (TOCIVID-19) (TOCIVID-19)". www.clinicaltrials.gov. National Library of Medicine. Retrieved 22 ខែមេសា 2020.
- ↑ "How doctors can potentially significantly reduce the number of deaths from Covid-19". Vox. 12 ខែមីនា 2020. Archived from the original on 19 ខែមីនា 2020. Retrieved 14 ខែមីនា 2020.
- ↑ (March 2020)"Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China". Intensive Care Medicine. DOI:10.1007/s00134-020-05991-x.
- ↑ Slater, Hannah (26 ខែមីនា 2020). "FDA Approves Phase III Clinical Trial of Tocilizumab for COVID-19 Pneumonia". www.cancernetwork.com. Cancer Network. Archived from the original on 17 ខែមិថុនា 2020. Retrieved 22 ខែមេសា 2020.
- ↑ (2017)"Preliminary Results of Prophylactic Tocilizumab after Axicabtageneciloleucel (axi-cel; KTE-C19) Treatment for Patients with Refractory,Aggressive Non-Hodgkin Lymphoma (NHL)". Blood 130 (Supplement 1). DOI:10.1182/blood.V130.Suppl_1.1547.1547.
- ↑ (2019)"GM-CSF inhibition reduces cytokine release syndrome and neuroinflammation but enhances CAR-T cell function in xenografts.". Blood. 133 (7): 697–709. DOI:10.1182/blood-2018-10-881722.
- ↑ "Northwell Health Initiates Clinical Trials of 2 COVID-19 Drugs". 21 March 2020. https://www.longislandpress.com/2020/03/21/northwell-health-initiates-clinical-trials-of-2-covid-19-drugs/.
- ↑ ៣៥០,០ ៣៥០,១ ៣៥០,២ ៣៥០,៣ (March 2020)"The convalescent sera option for containing COVID-19". The Journal of Clinical Investigation 130 (4): 1545–1548. DOI:10.1172/JCI138003.
- ↑ Pearce, Katie (13 ខែមីនា 2020). "Antibodies from COVID-19 survivors could be used to treat patients, protect those at risk: Infusions of antibody-laden blood have been used with reported success in prior outbreaks, including the SARS epidemic and the 1918 flu pandemic". The Hub at Johns Hopkins University. Archived from the original on 14 ខែមីនា 2020. Retrieved 14 ខែមីនា 2020.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help)
External links
[កែប្រែ]| ស្វែងយល់បន្ថែមអំពីជំងឺកូរ៉ូណាវីរុស២០១៩លើ គម្រោងបងប្អូនរបស់វិគីភីឌា: | |
| និយមន័យនិងបំណកប្រែពីវិគីនានុក្រម | |
| រូបភាពនិងមីឌាពី Wikimedia Commons | |
| ប្រភពរៀនសូត្រពីវិគីសាកលវិទ្យាល័យ | |
| រឿងរ៉ាវព័ត៌មានពីវិគីព័ត៌មាន | |
| សម្រង់ពាក្យសម្ដីពីវិគីសម្រង់ពាក្យ | |
| អត្ថបទប្រភពដើមពីវិគីប្រភព | |
Health agencies
[កែប្រែ]- Coronavirus disease (COVID-19) by the World Health Organization
- Coronavirus 2019 (COVID-19) by the U.S. Centers for Disease Control and Prevention
Directories
[កែប្រែ]Medical journals
[កែប្រែ]- Coronavirus Disease 2019 (COVID-19) by JAMA
- Coronavirus: News and Resources by the BMJ Publishing Group
- Novel Coronavirus Information Center by Elsevier
- COVID-19 Resource Centre by The Lancet
- SARS-CoV-2 and COVID-19 by Nature
- Coronavirus (Covid-19) by The New England Journal of Medicine
- Covid-19: Novel Coronavirus Archived 24 September 2020[Date mismatch] at the វេយប៊ែខ ម៉ាស៊ីន. by Wiley Publishing
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
ទំព័រគំរូ:Viral diseases ទំព័រគំរូ:COVID-19 pandemic
|
- Pages with reference errors
- CS1 errors: redundant parameter
- CS1 errors: unsupported parameter
- CS1 maint: url-status
- Pages containing cite templates with deprecated parameters
- CS1 កូរ៉េ-language sources (ko)
- CS1 អង់គ្លេស-language sources (en)
- អត្ថបទទាំងអស់ដែលមានតំណភ្ជាប់ក្រៅខូច
- អត្ថបទទាំងអស់ដែលមានតំណភ្ជាប់ក្រៅខូចពីApril 2020
- អត្ថបទដែលមានបរាមាត្រកាលបរិច្ឆេទអសុពលភាពក្នុងទំព័រគំរូ
- CS1 British English-language sources (en-gb)
- CS1 អាល្លឺម៉ង់-language sources (de)
- CS1 អ៊ីតាលី-language sources (it)
- អត្ថបទទាំងអស់ដែលមានតំណភ្ជាប់ក្រៅខូចពីតុលា 2022
- CS1 ព័រទុយហ្គាល់-language sources (pt)
- CS1 maint: numeric names: authors list
- CS1 American English-language sources (en-us)
- CS1 errors: deprecated parameters
- Pages using duplicate arguments in template calls
- អត្ថបទ with short description
- Short description is different from Wikidata
- អត្ថបទវិគីភីឌាដែលត្រូវការបកប្រែ
- Wikipedia articles in need of updating
- All Wikipedia articles in need of updating
- ប្រើប្រាស់កាលបរិច្ឆេទថ្ងៃ-ខែ-ឆ្នាំ-ពីMarch 2020
- Articles with hatnote templates targeting a nonexistent page
- Articles containing potentially dated statements from មេសា 2020
- Articles with invalid date parameter in template
- All articles containing potentially dated statements
- Pages using multiple image with auto scaled images
- អត្ថបទទាំងអស់ជាមួយការថ្លែងគ្មានប្រភព
- អត្ថបទជាមួយការថ្លែងគ្មានប្រភពពីMay 2020
- Articles lacking reliable referencesពីMarch 2020
- All articles lacking reliable references
- តំណវេយប៊ែខទំព័រគំរូវេបបណ្ណសារ
- Webarchive template warnings
- អត្ថបទវិគីភីឌាមានសញ្ញាណករល.គ.ប.ស
- អត្ថបទវិគីភីឌាមានសញ្ញាណករស.ន.ស
- អត្ថបទវិគីភីឌាមានសញ្ញាណករគ.ជ.ប
- COVID-19
- Occupational safety and health
- Viral respiratory tract infections
- Zoonoses
- ជំងឺបង្ករោគ